Stretchable, Self-Healing, and Bioactive Hydrogel with High-Functionality N,N'-bis(acryloyl)cystamine Dynamically Bonded Ag@polydopamine Crosslinkers for Wearable Sensors
- PMID: 39031305
- PMCID: PMC11425271
- DOI: 10.1002/advs.202404451
Stretchable, Self-Healing, and Bioactive Hydrogel with High-Functionality N,N'-bis(acryloyl)cystamine Dynamically Bonded Ag@polydopamine Crosslinkers for Wearable Sensors
Abstract
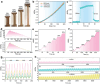
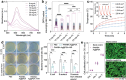
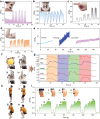
Hydrogels present attractive opportunities as flexible sensors due to their soft nature and tunable physicochemical properties. Despite significant advances, practical application of hydrogel-based sensor is limited by the lack of general routes to fabricate materials with combination of mechanical, conductive, and biological properties. Here, a multi-functional hydrogel sensor is reported by in situ polymerizing of acrylamide (AM) with N,N'-bis(acryloyl)cystamine (BA) dynamic crosslinked silver-modified polydopamine (PDA) nanoparticles, namely PAM/BA-Ag@PDA. Compared with traditional polyacrylamide (PAM) hydrogel, the BA-Ag@PDA nanoparticles provide both high-functionality crosslinks and multiple interactions within PAM networks, thereby endowing the optimized PAM/BA-Ag@PDA hydrogel with significantly enhanced tensile/compressive strength (349.80 kPa at 383.57% tensile strain, 263.08 kPa at 90% compressive strain), lower hysteresis (5.2%), improved conductivity (2.51 S m-1) and excellent near-infrared (NIR) light-triggered self-healing ability. As a strain sensor, the PAM/BA-Ag@PDA hydrogel shows a good sensitivity (gauge factor of 1.86), rapid response time (138 ms), and high stability. Owing to abundant reactive groups in PDA, the PAM/BA-Ag@PDA hydrogel exhibits inherent tissue adhesiveness and antioxidant, along with a synergistic antibacterial effect by PDA and Ag. Toward practical applications, the PAM/BA-Ag@PDA hydrogel can conformally adhere to skin and monitor subtle activities and large-scale movements with excellent reliability, demonstrating its promising applications as wearable sensors for healthcare.
Keywords: health monitoring; hydrogel; self‐healing; sensor.
© 2024 The Author(s). Advanced Science published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Zarei M., Lee G., Lee S. G., Cho K., Adv. Mater. 2023, 35, 2203193. - PubMed
-
- Xie C., Wang X., He H., Ding Y., Lu X., Adv. Funct. Mater. 2020, 30, 1909954.
-
- Liu D., Huyan C., Wang Z., Guo Z., Zhang X., Torun H., Mulvihill D., Xu B. B., Chen F., Mater. Horiz. 2023, 10, 2800. - PubMed
-
- Zhu T., Ni Y., Biesold G. M., Cheng Y., Ge M., Li H., Huang J., Lin Z., Lai Y., Chem. Soc. Rev. 2023, 52, 473. - PubMed
MeSH terms
Substances
Grants and funding
- 32071346/National Natural Science Foundation of China
- 32271403/National Natural Science Foundation of China
- 2021364/Youth Innovation Promotion Association, Chinese Academy of Sciences
- 2024A1515010223/GuangDong Basic and Applied Basic Research Foundation
- 2022A0505090007/Science and Technology Program of Guangdong Province
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
