Free serum concentrations of antibiotics determined by ultrafiltration: extensive evaluation of experimental variables
- PMID: 39041640
- PMCID: PMC11389746
- DOI: 10.1080/17576180.2024.2365526
Free serum concentrations of antibiotics determined by ultrafiltration: extensive evaluation of experimental variables
Abstract
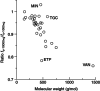
Aim: To assess the impact of experimental conditions on free serum concentrations as determined by ultrafiltration and HPLC-DAD analysis in a wide range of antibiotics.Materials & methods: Relative centrifugation force (RCF), temperature, pH and buffer were varied and the results compared with the standard protocol (phosphate buffer pH 7.4, 37°C, 1000 × g).Results: Generally, at 10,000 × g the unbound fraction (fu) decreased with increasing molecular weight, and was lower at 22°C. In unbuffered serum, the fu of flucloxacillin or valproic acid was increased, that of basic or amphoteric drugs considerably decreased. Comparable results were obtained using phosphate or HEPES buffer except for drugs which form metal chelate complexes.Conclusion: Maintaining a physiological pH is more important than strictly maintaining body temperature.
Keywords: HPLC-UV; anti-infectives; azole antimycotics; beta-lactams; plasma protein binding; therapeutic drug monitoring; unbound fraction; valproic acid.
Plain language summary
[Box: see text].
Conflict of interest statement
The authors have no competing interests or relevant affiliations with any organization or entity with an interest in or conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending or royalties.
Figures




Similar articles
-
Computerized advice on drug dosage to improve prescribing practice.Cochrane Database Syst Rev. 2013 Nov 12;2013(11):CD002894. doi: 10.1002/14651858.CD002894.pub3. Cochrane Database Syst Rev. 2013. PMID: 24218045 Free PMC article.
-
First-in-Human Predictions of Hepatic Clearance for Drugs With the Well-Stirred Model: Comparative Assessment Between Models of Fraction Unbound Based Either on the Free Drug Hypothesis, Albumin-Facilitated Hepatic Uptake or Dynamic Binding Kinetics.J Pharm Sci. 2024 Aug;113(8):2641-2650. doi: 10.1016/j.xphs.2024.05.021. Epub 2024 May 24. J Pharm Sci. 2024. PMID: 38796154
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
-
Antibiotic treatment for non-tuberculous mycobacteria lung infection in people with cystic fibrosis.Cochrane Database Syst Rev. 2025 Mar 27;3(3):CD016039. doi: 10.1002/14651858.CD016039. Cochrane Database Syst Rev. 2025. PMID: 40145528
-
[Fast determination of per- and polyfluoroalkyl substances in human serum by cold-induced phase separation coupled with liquid chromatography-tandem mass spectrometry].Se Pu. 2025 Jul;43(7):756-766. doi: 10.3724/SP.J.1123.2024.11028. Se Pu. 2025. PMID: 40610770 Free PMC article. Chinese.
Cited by
-
Toxicokinetics and analytical toxicology of the phenmetrazine-derived new psychoactive substance 3,4-methylenedioxyphenmetrazine studied by means of in vitro systems.Arch Toxicol. 2025 Apr;99(4):1407-1416. doi: 10.1007/s00204-025-03965-w. Epub 2025 Feb 4. Arch Toxicol. 2025. PMID: 39903277 Free PMC article.
-
Impact of Hypothermic Temperature Control on Plasma and Soft Tissue Pharmacokinetics of Penicillin/Beta-Lactamase Inhibitor Combinations in Patients Resuscitated After Cardiac Arrest.Clin Pharmacokinet. 2025 May;64(5):691-701. doi: 10.1007/s40262-025-01497-1. Epub 2025 Apr 10. Clin Pharmacokinet. 2025. PMID: 40208479 Free PMC article.
-
Target-site cefiderocol pharmacokinetics in soft tissues of healthy volunteers.J Antimicrob Chemother. 2024 Dec 2;79(12):3281-3288. doi: 10.1093/jac/dkae359. J Antimicrob Chemother. 2024. PMID: 39373642 Free PMC article.
References
-
- Kunin CM, Craig WA, Kornguth M, et al. Influence of binding on the pharmacologic activity of antibiotics. Ann NY Acad Sci. 1973;226:214–224. doi: 10.1111/j.1749-6632.1973.tb20483.x - DOI - PubMed
-
• Demonstrates the close relationship between the amount of free (unbound) drug and biological activity.
-
- Mouton JW, Dudley MN, Cars O, et al. Standardization of pharmacokinetic/pharmacodynamic (PK/PD) terminology for anti-infective drugs: an update. J Antimicrob Chemother. 2005;55:601–607. doi: 10.1093/jac/dki079 - DOI - PubMed
-
• Emphasizes the importance to refer all PK/PD indices to the free concentrations.
-
- European Medicines Agency (EMA) . Guideline on the use of pharmacokinetics and pharmacodynamics in the development of antimicrobial medicinal products [Internet]. 2016. Available from: https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-us...
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
