Real-world evidence of durable multi-dimensional improvement after 60-day peripheral nerve stimulation treatment used for shoulder pain
- PMID: 39041738
- PMCID: PMC11486131
- DOI: 10.1080/17581869.2024.2371779
Real-world evidence of durable multi-dimensional improvement after 60-day peripheral nerve stimulation treatment used for shoulder pain
Abstract
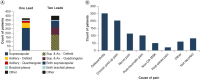
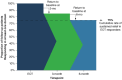
Aim: This real-world analysis aims to quantify improvements in multiple health domains in patients who received 60-day peripheral nerve stimulation (PNS) for shoulder pain.Materials & methods: Patients reported percent pain relief and Patient Global Impression of Change in quality of life, physical function and sleep at the end of treatment (EOT), 3 months, and 6 months.Results: Of 768 patients, 80.7% were responders in at least one domain at EOT. In a subset who were followed up, a cumulative 75% continued to respond in at least one domain through 6 months (85% [n = 140/165] at 3 months and 88% [n = 53/60] at 6 months).Conclusion: 60-day PNS used for shoulder pain produced multi-dimensional improvements across health domains at EOT and through 6 months.
Keywords: chronic pain; neuromodulation; pain management; peripheral nerve stimulation; real world data; shoulder pain.
Plain language summary
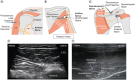
Chronic shoulder pain is common. Patients often have challenges with quality of life, physical function and sleep. Peripheral nerve stimulation (PNS) is one available treatment for chronic shoulder pain. Studies like this one show how treatments work in everyday use. These studies are important to help patients and doctors make decisions about their treatment. This study looked at patients who received a 60-day PNS treatment for their shoulder pain. The treatment involved one or two small wires placed through the skin to stimulate nerves in the shoulder. At the end of treatment, they reported how much pain relief they had. They also reported how much their quality of life, physical function and sleep changed. Of 768 patients, most (80.7%) had improvement in at least one of these areas at the end of their PNS treatment. Only 5.9% of patients reported complications. The most common complaint was skin irritation. A smaller group of patients also shared how they were doing 3 months and 6 months after starting treatment. Overall, about 75% of patients continued to have improvement in at least one area 6 months after starting treatment. For many people, PNS provided improvements in pain and in other areas important to patients. These results suggest that 60-day PNS can be a valuable treatment for people with shoulder pain.
Conflict of interest statement
A Valimahomed has received consultancy and speaker fees from Nalu, consultancy fees from Vertos and consultancy fees, speaker fees and research funding from SPR Therapeutics. D Dickerson has received consultancy fees from Biotronik, Nalu, and Pfizer; consultancy and speaker fees from Vertos; consultancy fees and research funding from SPR Therapeutics; and speaker fees, consultancy fees, and research funding from Abbott. H Vucetic has received consultancy and speaker fees from Vertos, Boston Scientific, Saluda Medical, Intrinsic Therapeutics and SPR Therapeutics. J Rutledge has received consultancy fees from Boston Scientific and TerSera, and consultancy and speaker fees from Biotronik and SPR Therapeutics. CAZ is an employee of SPR Therapeutics and is a coinventor on patents related to the technology analyzed herein. ND Crosby and JW Boggs are employees of SPR Therapeutics, are coinventors on patents related to the technology analyzed herein, and hold stock options for SPR Therapeutics. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
Figures

References
MeSH terms
LinkOut - more resources
Full Text Sources
