The metabolic role of vitamin D in children's neurodevelopment: a network study
- PMID: 39043876
- PMCID: PMC11266698
- DOI: 10.1038/s41598-024-67835-8
The metabolic role of vitamin D in children's neurodevelopment: a network study
Abstract
Neurodevelopmental disorders are rapidly increasing in prevalence and have been linked to various environmental risk factors. Mounting evidence suggests a potential role of vitamin D in child neurodevelopment, though the causal mechanisms remain largely unknown. Here, we investigate how vitamin D deficiency affects children's communication development, particularly in relation to Autism Spectrum Disorder (ASD). We do so by developing an integrative network approach that combines metabolomic profiles, clinical traits, and neurodevelopmental data from a pediatric cohort. Our results show that low levels of vitamin D are associated with changes in the metabolic networks of tryptophan, linoleic, and fatty acid metabolism. These changes correlate with distinct ASD-related phenotypes, including delayed communication skills and respiratory dysfunctions. Additionally, our analysis suggests the kynurenine and serotonin sub-pathways may mediate the effect of vitamin D on early life communication development. Altogether, our findings provide metabolome-wide insights into the potential of vitamin D as a therapeutic option for ASD and other communication disorders.
© 2024. The Author(s).
Conflict of interest statement
S.T.W. receives royalties from UpToDate and is on the board of Histolix, a digital pathology company. All other authors declare that they have no competing interests.
Figures


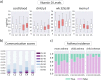

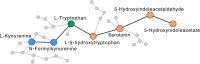
Update of
-
The metabolic role of vitamin D in children's neurodevelopment: a network study.bioRxiv [Preprint]. 2023 Jul 15:2023.06.23.546277. doi: 10.1101/2023.06.23.546277. bioRxiv. 2023. Update in: Sci Rep. 2024 Jul 23;14(1):16929. doi: 10.1038/s41598-024-67835-8. PMID: 37425858 Free PMC article. Updated. Preprint.
References
-
- Data, C. statistics on autism spectrum disorder|CDC. Centers Dis. Control Prevent.2020, 4781–4792 (2020).
MeSH terms
Substances
Grants and funding
- R01HL091528/U.S. Department of Health & Human Services | NIH | National Heart, Lung, and Blood Institute (NHLBI)
- K25 HL168157/HL/NHLBI NIH HHS/United States
- K01HL146980/U.S. Department of Health & Human Services | NIH | National Heart, Lung, and Blood Institute (NHLBI)
- R01HL155749/U.S. Department of Health & Human Services | NIH | National Heart, Lung, and Blood Institute (NHLBI)
- R01HL123915/U.S. Department of Health & Human Services | NIH | National Heart, Lung, and Blood Institute (NHLBI)
- UH3 OD023268/OD/NIH HHS/United States
- K25HL168157/U.S. Department of Health & Human Services | NIH | National Heart, Lung, and Blood Institute (NHLBI)
- R01 HL155749/HL/NHLBI NIH HHS/United States
- K01HL153941/U.S. Department of Health & Human Services | NIH | National Heart, Lung, and Blood Institute (NHLBI)
- K01 HL153941/HL/NHLBI NIH HHS/United States
- R01HL141826/U.S. Department of Health & Human Services | NIH | National Heart, Lung, and Blood Institute (NHLBI)
- UH3 OD023268/CD/ODCDC CDC HHS/United States
- P30 ES001247/ES/NIEHS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical

