Neoadjuvant anthracycline followed by toripalimab combined with nab-paclitaxel in patients with early triple-negative breast cancer (NeoTENNIS): a single-arm, phase II study
- PMID: 39045544
- PMCID: PMC11260571
- DOI: 10.1016/j.eclinm.2024.102700
Neoadjuvant anthracycline followed by toripalimab combined with nab-paclitaxel in patients with early triple-negative breast cancer (NeoTENNIS): a single-arm, phase II study
Abstract
Background: Toripalimab, a novel PD-1 antibody, is approved for treatment of multiple solid tumors; however, its neoadjuvant use with chemotherapy for triple-negative breast cancer (TNBC) remains unevaluated. Additionally, induction chemotherapy followed by de-escalation of neoadjuvant immunotherapy remains underexplored. Therefore, we conducted a phase II trial investigating a novel neoadjuvant chemoimmunotherapy regimen including de-escalation of immunotherapy for early-stage TNBC.
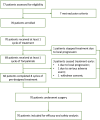
Methods: Chemotherapy and anti-PD-1 therapy were sequentially administered in a neoadjuvant setting to female patients with histologically confirmed stage II-III TNBC between June 9, 2020, and March 24, 2022. Patients received neoadjuvant therapy with four cycles of epirubicin-cyclophosphamide every 2 weeks, followed by toripalimab (240 mg) every 3 weeks plus nab-paclitaxel weekly for 12 weeks. The primary endpoint was total pathological complete response (tpCR; ypT0/is ypN0). Key secondary endpoints included breast pCR (bpCR; ypT0/is), event-free survival and biomarker analysis. Safety was also assessed. This study was registered with ClinicalTrials.gov (NCT04418154).
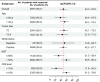
Findings: Among 70 enrolled patients (median age, 51 years; 62.9% stage III), 66 completed treatment without progression and subsequently underwent surgery. The percentages of patients with a tpCR and bpCR were 39 of 70 (55.7%, 95% confidence interval [CI]: 43.3-67.6) and 41 of 70 (58.6%, 95% CI 46.2-70.2), respectively. Sixteen (22.9%) patients experienced grade ≥3 adverse events (AEs), frequently neutropenia (12, 17.1%) and leukopenia (11, 15.7%). The most common immune-related AE was hypothyroidism (5, 7.1%, all grade 1-2).
Interpretation: Including 12 weeks of toripalimab in neoadjuvant chemotherapy conferred encouraging activity and manageable toxicity in patients with early TNBC, and this regimen warrants further investigation.
Funding: National Natural Science Foundation of China, Junshi Biosciences, and Jiangsu Hengrui Pharmaceuticals.
Keywords: De-escalation; Immune checkpoint inhibitor; Induction treatment; Neoadjuvant therapy; Triple-negative breast cancer.
© 2024 Published by Elsevier Ltd.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. Funding sources were not involved in the study design, data collection, analysis and interpretation, writing of the report, or decision to submit the article for publication.
Figures

References
-
- Mougalian S.S., Soulos P.R., Killelea B.K., et al. Use of neoadjuvant chemotherapy for patients with stage I to III breast cancer in the United States. Cancer. 2015;121:2544–2552. - PubMed
-
- Cortazar P., Zhang L., Untch M., et al. Pathological complete response and long-term clinical benefit in breast cancer: the CTNeoBC pooled analysis. Lancet. 2014;384:164–172. - PubMed
-
- Schmid P., Rugo H.S., Adams S., et al. Atezolizumab plus nab-paclitaxel as first-line treatment for unresectable, locally advanced or metastatic triple-negative breast cancer (IMpassion 130): updated efficacy results from a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2020;21:44–59. - PubMed
-
- Schmid P., Cortes J., Pusztai L., et al. Pembrolizumab for early triple-negative. Breast Cancer. 2020;382:810–821. - PubMed
Associated data
LinkOut - more resources
Full Text Sources
Medical

