Investigation of Peroxisome Proliferator-Activated Receptor Genes as Requirements for Visual Startle Response Hyperactivity in Larval Zebrafish Exposed to Structurally Similar Per- and Polyfluoroalkyl Substances (PFAS)
- PMID: 39046251
- PMCID: PMC11268134
- DOI: 10.1289/EHP13667
Investigation of Peroxisome Proliferator-Activated Receptor Genes as Requirements for Visual Startle Response Hyperactivity in Larval Zebrafish Exposed to Structurally Similar Per- and Polyfluoroalkyl Substances (PFAS)
Abstract
Background: Per- and polyfluoroalkyl Substances (PFAS) are synthetic chemicals widely detected in humans and the environment. Exposure to perfluorooctanesulfonic acid (PFOS) or perfluorohexanesulfonic acid (PFHxS) was previously shown to cause dark-phase hyperactivity in larval zebrafish.
Objectives: The objective of this study was to elucidate the mechanism by which PFOS or PFHxS exposure caused hyperactivity in larval zebrafish.
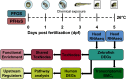
Methods: Swimming behavior was assessed in 5-d postfertilization (dpf) larvae following developmental (1-4 dpf) or acute (5 dpf) exposure to PFOS, PFHxS, or 0.4% dimethyl sulfoxide (DMSO). After developmental exposure and chemical washout at 4 dpf, behavior was also assessed at 5-8 dpf. RNA sequencing was used to identify differences in global gene expression to perform transcriptomic benchmark concentration-response () modeling, and predict upstream regulators in PFOS- or PFHxS-exposed larvae. CRISPR/Cas9-based gene editing was used to knockdown peroxisome proliferator-activated receptors (ppars) pparaa/ab, pparda/db, or pparg at day 0. Knockdown crispants were exposed to PFOS or 0.4% DMSO from 1-4 dpf and behavior was assessed at 5 dpf. Coexposure with the ppard antagonist GSK3787 and PFOS was also performed.
Results: Transient dark-phase hyperactivity occurred following developmental or acute exposure to PFOS or PFHxS, relative to the DMSO control. In contrast, visual startle response (VSR) hyperactivity only occurred following developmental exposure and was irreversible up to 8 dpf. Similar global transcriptomic profiles, estimates, and enriched functions were observed in PFOS- and PFHxS-exposed larvae, and ppars were identified as putative upstream regulators. Knockdown of pparda/db, but not pparaa/ab or pparg, blunted PFOS-dependent VSR hyperactivity to control levels. This finding was confirmed via antagonism of ppard in PFOS-exposed larvae.
Discussion: This work identifies a novel adverse outcome pathway for VSR hyperactivity in larval zebrafish. We demonstrate that developmental, but not acute, exposure to PFOS triggered persistent VSR hyperactivity that required ppard function. https://doi.org/10.1289/EHP13667.
Figures


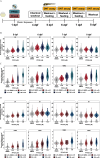


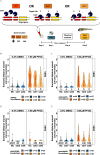
![Figure 8 is one violin plot and one table. The violin plot titled Visual startle response 2, plotting Summed distance moved (centimeters per 3 seconds), ranging from 0 to 5 in unit increments (y-axis) across Dimethyl sulfoxide with 4-Chloro-N-(2-{[5-trifluoromethyl)-2-pyridyl]sulfonyl}ethyl)benzamide, Perfluorooctanesulfonic acid with 4-Chloro-N-(2-{[5-trifluoromethyl)-2-pyridyl]sulfonyl}ethyl)benzamide, and Perfluorooctanesulfonic acid with 4-Chloro-N-(2-{[5-trifluoromethyl)-2-pyridyl]sulfonyl}ethyl)benzamide (x-axis). A tabular representation, in three columns, lists 4-Chloro-N-(2-{[5-trifluoromethyl)-2-pyridyl]sulfonyl}ethyl)benzamide, Perfluorooctanesulfonic acid (micromolar), and number of cases. Row 1: 0, 0, 55 cases. Row 2: 9.25, 0, and 28 cases. Row 3: 27.2, 0, and 29 cases. Row 4: 0, 7.86, and 49 cases. Row 5: 9.25, 7.86, and 33 cases. Row 6: 27.2, 7.86, and 19 cases. Row 7: 0, 11.72, and 33 cases. Row 8: 9.25, 11.72, and 18 cases. Row 9: 27.2, 11.72, and 16 cases.](https://cdn.ncbi.nlm.nih.gov/pmc/blobs/b349/11268134/04dfe2cbb3d7/ehp13667_f8.gif)
References
-
- Banks RE, Smart BE, Tatlow J. 2013. Organofluorine Chemistry: Principles and Commercial Applications. New York, NY: Springer Science & Business Media.
-
- Kissa E. 2001. Fluorinated Surfactants and Repellents. 2nd ed. New York, NY: Marcel Dekker.
-
- Schultz MM, Barofsky DF, Field JA. 2003. Fluorinated alkyl surfactants. Environ Eng Sci 20(5):487–501, 10.1089/109287503768335959. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

