Role of STAT3-FOXO3 Signaling in the Modulation of Neuroplasticity by PD-L1-HGF-Decorated Mesenchymal Stem Cell-Derived Exosomes in a Murine Stroke Model
- PMID: 39049677
- PMCID: PMC11423231
- DOI: 10.1002/advs.202404882
Role of STAT3-FOXO3 Signaling in the Modulation of Neuroplasticity by PD-L1-HGF-Decorated Mesenchymal Stem Cell-Derived Exosomes in a Murine Stroke Model
Abstract
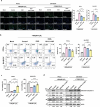
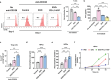
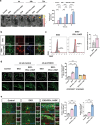
The limited therapeutic strategies available for stroke leave many patients disabled for life. This study assessed the potential of programmed death-ligand 1 (PD-L1) and hepatocyte growth factor (HGF)-engineered mesenchymal stem cell-derived exosomes (EXO-PD-L1-HGF) in enhancing neurological recovery post-stroke. EXO-PD-L1-HGF, which efficiently endocytosed into target cells, significantly diminishes the H2O2-induced neurotoxicity and increased the antiapoptotic proteins in vitro. EXO-PD-L1-HGF attenuates inflammation by inhibiting T-cell proliferation and increasing the number of CD8+CD122+IL-10+ regulatory T cells. Intravenous injection of EXO-PD-L1-HGF could target stromal cell-derived factor-1α (SDF-1α+) cells over the peri-infarcted area of the ischemic brain through CXCR4 upregulation and accumulation in neuroglial cells post-stroke. EXO-PD-L1-HGF facilitates endogenous nestin+ neural progenitor cell (NPC)-induced neurogenesis via STAT3-FOXO3 signaling cascade, which plays a pivotal role in cell survival and neuroprotection, thereby mitigating infarct size and enhancing neurological recovery in a murine stroke model. Moreover, increasing populations of the immune-regulatory CD19+IL-10+ and CD8+CD122+IL-10+ cells, together with reducing populations of proinflammatory cells, created an anti-inflammatory microenvironment in the ischemic brain. Thus, innovative approaches employing EXO-PD-L1-HGF intervention, which targets SDF-1α+ expression, modulates the immune system, and enhances the activation of resident nestin+ NPCs, might significantly alter the brain microenvironment and create a niche conducive to inducing neuroplastic regeneration post-stroke.
Keywords: HGF; PD‐L1; STAT3‐FOXO3 pathway; exosomes; stroke.
© 2024 The Author(s). Advanced Science published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





References
-
- Lackland D. T., Roccella E. J., Deutsch A. F., Fornage M., George M. G., Howard G., Kissela B. M., Kittner S. J., Lichtman J. H., Lisabeth L. D., Schwamm L. H., Smith E. E., Towfighi A., C. American Heart Association Stroke , C. Council on , N. Stroke , C. Council on Quality of , R. Outcomes , G. Council on Functional , B. Translational , Stroke 2014, 45, 315. - PMC - PubMed
-
- a) Potla N., Ganti L., Int. J. Emerg. Med. 2022, 15, 1; - PMC - PubMed
- b) Jovin T. G., Nogueira R. G., Lansberg M. G., Demchuk A. M., Martins S. O., Mocco J., Ribo M., Jadhav A. P., Ortega‐Gutierrez S., Hill M. D., Lima F. O., Haussen D. C., Brown S., Goyal M., Siddiqui A. H., Heit J. J., Menon B. K., Kemp S., Budzik R., Urra X., Marks M. P., Costalat V., Liebeskind D. S., Albers G. W., Lancet 2022, 399, 249. - PubMed
-
- Zivin J. A., Ann. Neurol. 2009, 66, 6.
-
- Lee J., Chang W. H., Chung J. W., Kim S. J., Kim S. K., Lee J. S., Sohn S. I., Kim Y. H., Bang O. Y., Collaborators S., Stroke 2022, 53, 20.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
