The Spectrum of CAR Cellular Effectors: Modes of Action in Anti-Tumor Immunity
- PMID: 39061247
- PMCID: PMC11274444
- DOI: 10.3390/cancers16142608
The Spectrum of CAR Cellular Effectors: Modes of Action in Anti-Tumor Immunity
Abstract
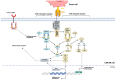
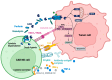
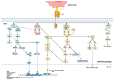
Chimeric antigen receptor-T cells have spearheaded the field of adoptive cell therapy and have shown remarkable results in treating hematological neoplasia. Because of the different biology of solid tumors compared to hematological tumors, response rates of CAR-T cells could not be transferred to solid entities yet. CAR engineering has added co-stimulatory domains, transgenic cytokines and switch receptors to improve performance and persistence in a hostile tumor microenvironment, but because of the inherent cell type limitations of CAR-T cells, including HLA incompatibility, toxicities (cytokine release syndrome, neurotoxicity) and high costs due to the logistically challenging preparation process for autologous cells, the use of alternative immune cells is gaining traction. NK cells and γδ T cells that do not need HLA compatibility or macrophages and dendritic cells with additional properties such as phagocytosis or antigen presentation are increasingly seen as cellular vehicles with potential for application. As these cells possess distinct properties, clinicians and researchers need a thorough understanding of their peculiarities and commonalities. This review will compare these different cell types and their specific modes of action seen upon CAR activation.
Keywords: adoptive cell therapy; chemokines; chimeric antigen receptor; cytokines; dendritic cells; gamma-delta T cells; killing mechanism; kinetics; macrophages; mode of action; natural killer cells; persistence.
Conflict of interest statement
S.K. has received honoraria from Cymab, Plectonic, TCR2 Inc., Miltenyi, Galapagos, Novartis, BMS and GSK. S.K. is an inventor of several patents in the field of immuno-oncology. S.K. received license fees from TCR2 Inc and Carina Biotech. S.K. received research support from TCR2 Inc., Tabby Therapeutics, Catalym GmBH, Plectonic GmBH and Arcus Bioscience for work unrelated to the manuscript. M.H. is listed an inventor on patent applications and granted patents related to CAR technology, licensed in part to industry. Co-founder and equity owner T-CURX GmbH, Würzburg. Speaker honoraria: BMS, Janssen, Kite/Gilead, Novartis. Research support: BMS. J.W. is listed as an inventor on patent applications and granted patents related to CAR technology. J.F. is listed an inventor on patent applications and granted patents related to CAR technology, licensed in part to industry.
Figures



References
-
- Prehn R.T., Main J.M. Immunity to Methylcholanthrene-Induced Sarcomas. J. Natl. Cancer Inst. 1957;18:769–778. - PubMed
-
- Klein G., Sjogren H.O., Klein E., Hellstrom K.E. Demonstration of Resistance against Methylcholanthrene-Induced Sarcomas in the Primary Autochthonous Host. Cancer Res. 1960;20:1561–1572. - PubMed
-
- Rosenberg S.A., Packard B.S., Aebersold P.M., Solomon D., Topalian S.L., Toy S.T., Simon P., Lotze M.T., Yang J.C., Seipp C.A., et al. Use of Tumor-Infiltrating Lymphocytes and Interleukin-2 in the Immunotherapy of Patients with Metastatic Melanoma. N. Engl. J. Med. 1988;319:1676–1680. doi: 10.1056/NEJM198812223192527. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials

