PGC1-Alpha/Sirt3 Signaling Pathway Mediates the Anti-Pulmonary Fibrosis Effect of Hirudin by Inhibiting Fibroblast Senescence
- PMID: 39062010
- PMCID: PMC11274105
- DOI: 10.3390/biomedicines12071436
PGC1-Alpha/Sirt3 Signaling Pathway Mediates the Anti-Pulmonary Fibrosis Effect of Hirudin by Inhibiting Fibroblast Senescence
Abstract
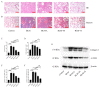
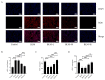
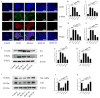
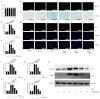
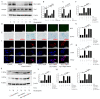
Idiopathic pulmonary fibrosis (IPF) is a chronic, progressive fibrotic lung disease for which there is a lack of effective pharmacological treatments. Hirudin, a natural peptide extracted from leeches, has been used for broad pharmacological purposes. In this study, we investigated the therapeutic effects of hirudin on IPF and its related mechanism of action. By constructing a mouse model of pulmonary fibrosis and treating it with hirudin in vivo, we found that hirudin exerted anti-fibrotic, anti-oxidative, and anti-fibroblast senescence effects. Moreover, using an in vitro model of stress-induced premature senescence in primary mouse lung fibroblasts and treating with hirudin, we observed inhibition of fibroblast senescence and upregulation of PGC1-alpha and Sirt3 expression. However, specific silencing of PGC1-alpha or Sirt3 suppressed the anti-fibroblast senescence effect of hirudin. Thus, the PGC1-alpha/Sirt3 pathway mediates the anti-fibroblast senescence effect of hirudin, potentially serving as a molecular mechanism underlying its anti-fibrosis and anti-oxidative stress effects exerted on the lungs.
Keywords: PGC1-alpha/Sirt3 pathway; fibroblast senescence; hirudin; idiopathic pulmonary fibrosis.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Hesperidin inhibits lung fibroblast senescence via IL-6/STAT3 signaling pathway to suppress pulmonary fibrosis.Phytomedicine. 2023 Apr;112:154680. doi: 10.1016/j.phymed.2023.154680. Epub 2023 Jan 29. Phytomedicine. 2023. PMID: 36736168
-
Baicalein attenuates bleomycin-induced lung fibroblast senescence and lung fibrosis through restoration of Sirt3 expression.Pharm Biol. 2023 Dec;61(1):288-297. doi: 10.1080/13880209.2022.2160767. Pharm Biol. 2023. PMID: 36815239 Free PMC article.
-
Therapeutic effects of the rhSOD2-Hirudin fusion protein on bleomycin-induced pulmonary fibrosis in mice.Eur J Pharmacol. 2019 Jun 5;852:77-89. doi: 10.1016/j.ejphar.2019.03.001. Epub 2019 Mar 2. Eur J Pharmacol. 2019. PMID: 30831079
-
Fibroblast senescence in the pathology of idiopathic pulmonary fibrosis.Am J Physiol Lung Cell Mol Physiol. 2018 Aug 1;315(2):L162-L172. doi: 10.1152/ajplung.00037.2018. Epub 2018 Apr 26. Am J Physiol Lung Cell Mol Physiol. 2018. PMID: 29696986 Free PMC article. Review.
-
Cell senescence and fibrotic lung diseases.Exp Gerontol. 2020 Apr;132:110836. doi: 10.1016/j.exger.2020.110836. Epub 2020 Jan 17. Exp Gerontol. 2020. PMID: 31958492 Free PMC article. Review.
Cited by
-
SIRT3/6/7: promising therapeutic targets for pulmonary fibrosis.Front Cell Dev Biol. 2025 Apr 2;13:1557384. doi: 10.3389/fcell.2025.1557384. eCollection 2025. Front Cell Dev Biol. 2025. PMID: 40241794 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources

