Third-Generation Tetracyclines: Current Knowledge and Therapeutic Potential
- PMID: 39062497
- PMCID: PMC11275049
- DOI: 10.3390/biom14070783
Third-Generation Tetracyclines: Current Knowledge and Therapeutic Potential
Abstract
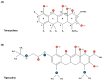
Tetracyclines constitute a unique class of antibiotic agents, widely prescribed for both community and hospital infections due to their broad spectrum of activity. Acting by disrupting protein synthesis through tight binding to the 30S ribosomal subunit, their interference is typically reversible, rendering them bacteriostatic in action. Resistance to tetracyclines has primarily been associated with changes in pump efflux or ribosomal protection mechanisms. To address this challenge, tetracycline molecules have been chemically modified, resulting in the development of third-generation tetracyclines. These novel tetracyclines offer significant advantages in treating infections, whether used alone or in combination therapies, especially in hospital settings. Beyond their conventional antimicrobial properties, research has highlighted their potential non-antibiotic properties, including their impact on immunomodulation and malignancy. This review will focus on third-generation tetracyclines, namely tigecycline, eravacycline, and omadacycline. We will delve into their mechanisms of action and resistance, while also evaluating their pros and cons over time. Additionally, we will explore their therapeutic potential, analyzing their primary indications of prescription, potential future uses, and non-antibiotic features. This review aims to provide valuable insights into the clinical applications of third-generation tetracyclines, thereby enhancing understanding and guiding optimal clinical use.
Keywords: eravacycline; immunomodulation; malignancy; mechanism of action; non-antibiotic properties; omadacycline; resistance; tetracyclines; tigecycline.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

