Auto/Paracrine C-Type Natriuretic Peptide/Cyclic GMP Signaling Prevents Endothelial Dysfunction
- PMID: 39063044
- PMCID: PMC11277478
- DOI: 10.3390/ijms25147800
Auto/Paracrine C-Type Natriuretic Peptide/Cyclic GMP Signaling Prevents Endothelial Dysfunction
Abstract
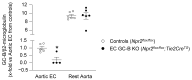
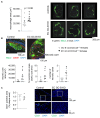
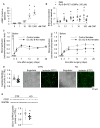
Endothelial dysfunction is cause and consequence of cardiovascular diseases. The endothelial hormone C-type natriuretic peptide (CNP) regulates vascular tone and the vascular barrier. Its cGMP-synthesizing guanylyl cyclase-B (GC-B) receptor is expressed in endothelial cells themselves. To characterize the role of endothelial CNP/cGMP signaling, we studied mice with endothelial-selective GC-B deletion. Endothelial EC GC-B KO mice had thicker, stiffer aortae and isolated systolic hypertension. This was associated with increased proinflammatory E-selectin and VCAM-1 expression and impaired nitric oxide bioavailability. Atherosclerosis susceptibility was evaluated in such KO and control littermates on Ldlr (low-density lipoprotein receptor)-deficient background fed a Western diet for 10 weeks. Notably, the plaque areas and heights within the aortic roots were markedly increased in the double EC GC-B/Ldlr KO mice. This was accompanied by enhanced macrophage infiltration and greater necrotic cores, indicating unstable plaques. Finally, we found that EC GC-B KO mice had diminished vascular regeneration after critical hind-limb ischemia. Remarkably, all these genotype-dependent changes were only observed in female and not in male mice. Auto/paracrine endothelial CNP/GC-B/cGMP signaling protects from arterial stiffness, systolic hypertension, and atherosclerosis and improves reparative angiogenesis. Interestingly, our data indicate a sex disparity in the connection of diminished CNP/GC-B activity to endothelial dysfunction.
Keywords: C-type natriuretic peptide; angiogenesis; arterial stiffening; atherosclerosis; cyclic GMP; endothelial dysfunction; systolic hypertension.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- Wu J., Thabet S.R., Kirabo A., Trott D.W., Saleh M.A., Xiao L., Madhur M.S., Chen W., Harrison D.G. Inflammation and mechanical stretch promote aortic stiffening in hypertension through activation of p38 mitogen-activated protein kinase. Circ. Res. 2014;114:616–625. doi: 10.1161/CIRCRESAHA.114.302157. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

