Recent Advances of Natural Pentacyclic Triterpenoids as Bioactive Delivery System for Synergetic Biological Applications
- PMID: 39063310
- PMCID: PMC11275325
- DOI: 10.3390/foods13142226
Recent Advances of Natural Pentacyclic Triterpenoids as Bioactive Delivery System for Synergetic Biological Applications
Abstract
Bioactive compounds have drawn much attention according to their various health benefits. However, poor dissolvability, low stability and limited bioavailability largely narrow their applications. Although a variety of nontoxic ingredients have been rapidly developed as vehicles to deliver bioactive compounds in the last few years, most of them are non-bioactive. Pentacyclic triterpenoids, owing to their unique self-assembly and co-assembly behaviors and different physiological functions, can construct bioactive carriers due to their higher biodegradability, biocompatibility and lower toxicity. In this paper, the basic classification, biological activities and physicochemical properties of pentacyclic triterpenoids were summarized. Additionally, applications of self-assembled and co-assembled pentacyclic triterpenoids as bioactive delivery systems to load bioactive components and future research directions were discussed. This study emphasizes the potential of pentacyclic triterpenoids as bioactive delivery systems, offering a new perspective for constructing self- or co-assemblies for further synergetic biological applications.
Keywords: bioactive nanocarriers; biological activity; natural small molecules; pentacyclic triterpenoids; self-assembly.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures

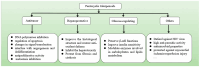
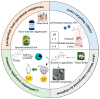
References
-
- Wani T.A., Shah A.G., Wani S.M., Wani I.A., Masoodi F.A., Nissar N., Shagoo M.A. Suitability of different food grade materials for the encapsulation of some functional foods well reported for their advantages and susceptibility. Crit. Rev. Food Sci. Nutr. 2016;56:2431–2454. doi: 10.1080/10408398.2013.845814. - DOI - PubMed
-
- Zhong H., Zhu Y., Yu J., Xu X., Gleeson J.P., Ryan S.M., Brayden D.J. Oral delivery strategies for nutraceuticals: Delivery vehicles and absorption enhancers. Trends Food Sci. Technol. 2016;53:90–101.
Publication types
Grants and funding
- No. SKLFNS-KF-202301/the Open Project Program of State Key Laboratory of Food Nutrition and Safety
- No. BTBUYXTD202202/Ministry of Science and Technology of China, the fund of Cultivation Project of Double First-Class Disciplines of Food Science and Engineering, Beijing Technology & Business University
LinkOut - more resources
Full Text Sources

