Influence of the Acceptor Fluid on the Bupivacaine Release from the Prospective Intra-Articular Methylcellulose Hydrogel
- PMID: 39065564
- PMCID: PMC11279645
- DOI: 10.3390/pharmaceutics16070867
Influence of the Acceptor Fluid on the Bupivacaine Release from the Prospective Intra-Articular Methylcellulose Hydrogel
Abstract
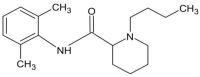
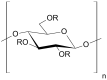
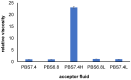
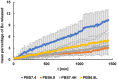
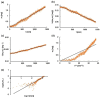
Injections are one way of delivering drugs directly to the joint capsule. Employing this possibility, local anesthetic, such as bupivacaine (Bu), in the form of the suspension can be administered. The aim of this work was to propose a methylcellulose-based hydrogel-incorporated bupivacaine for intra-articular injections and to study the release kinetics of the drug from the hydrogel to different acceptor media, reflecting the synovial fluid of a healthy joint and the synovial fluid of an inflamed joint. The drug release studies were performed employing the flow apparatus. The drug was released to four different acceptor fluids: phosphate buffer pH = 7.4 (PBS7.4), phosphate buffer pH = 6.8 (PBS6.8), phosphate buffer pH = 7.4 with the high-molecular-weight sodium hyaluronate (PBS7.4H), and phosphate buffer pH = 6.8 with the low-molecular-weight sodium hyaluronate (PBS6.8L). The investigation was carried out at the temperature of 37 °C. The absorbance of the Bu released was measured at the wavelength of 262 nm every 2 min for 24 h. The release profiles of Bu to the acceptor media PBS7.4, PBS6.8, PBS7.4H, and PBS6.8L were described best by the first-order kinetics and the second-order equation. According to these models, the release rate constants were the highest when Bu was released to the fluid PBS7.4 and were k1 = (7.20 ± 0.01) × 10-5 min-1 and k2 = (3.00 ± 0.04) × 10-6 mg-1 × min-1, respectively. The relative viscosity of the acceptor medium, its pH, and the addition of high-molecular-weight or low-molecular-weight sodium hyaluronate (HAH or HAL) to the acceptor fluid influenced the drug dissolution. The release of Bu into the medium reflecting healthy synovial fluid takes a different pattern from its release into the fluid of an inflamed joint.
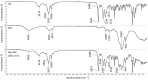
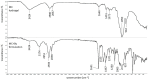
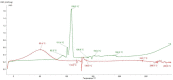
Keywords: DSC study; FTIR study; bupivacaine; intra-articular hydrogel; kinetics; release; sodium hyaluronate.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
The Effects of Synthetic Polymers on the Release Patterns of Bupivacaine Hydrochloride from Sodium Hyaluronate Hydrogels.Biomedicines. 2024 Dec 27;13(1):39. doi: 10.3390/biomedicines13010039. Biomedicines. 2024. PMID: 39857623 Free PMC article.
-
The Interactions and Release Kinetics of Sodium Hyaluronate Implemented in Nonionic and Anionic Polymeric Hydrogels, Studied by Immunoenzymatic ELISA Test.Pharmaceutics. 2021 Dec 27;14(1):58. doi: 10.3390/pharmaceutics14010058. Pharmaceutics. 2021. PMID: 35056954 Free PMC article.
-
Crystal suspensions of poorly soluble peptides for intra-articular application: a novel approach for biorelevant assessment of their in vitro release.Int J Pharm. 2014 Jan 30;461(1-2):46-53. doi: 10.1016/j.ijpharm.2013.11.031. Epub 2013 Nov 23. Int J Pharm. 2014. PMID: 24280019
-
Is chemical incompatibility responsible for chondrocyte death induced by local anesthetics?Am J Sports Med. 2010 Mar;38(3):520-6. doi: 10.1177/0363546509349799. Am J Sports Med. 2010. PMID: 20194957
-
Potential mechanism of action of intra-articular hyaluronan therapy in osteoarthritis: are the effects molecular weight dependent?Semin Arthritis Rheum. 2002 Aug;32(1):10-37. doi: 10.1053/sarh.2002.33720. Semin Arthritis Rheum. 2002. PMID: 12219318 Review.
Cited by
-
Advanced Hydrogel Systems for Local Anesthetic Delivery: Toward Prolonged and Targeted Pain Relief.Gels. 2025 Feb 12;11(2):131. doi: 10.3390/gels11020131. Gels. 2025. PMID: 39996674 Free PMC article. Review.
-
The Effects of Synthetic Polymers on the Release Patterns of Bupivacaine Hydrochloride from Sodium Hyaluronate Hydrogels.Biomedicines. 2024 Dec 27;13(1):39. doi: 10.3390/biomedicines13010039. Biomedicines. 2024. PMID: 39857623 Free PMC article.
References
-
- Temple-Wong M.M., Ren S., Quach P., Hansen B.C., Chen A.C., Hasegawa A., D’Lima D.D., Koziol J., Masuda K., Lotz M.K., et al. Hyaluronan concentration and size distribution in human knee synovial fluid: Variations with age and cartilage degeneration. Arthritis Res. Ther. 2016;18:18. doi: 10.1186/s13075-016-0922-4. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

