Effects of Postprandial Factors and Second Meal Intake Time on Bioequivalence Investigation of Tadalafil-Loaded Orodispersible Films in Human Volunteers
- PMID: 39065611
- PMCID: PMC11280306
- DOI: 10.3390/pharmaceutics16070915
Effects of Postprandial Factors and Second Meal Intake Time on Bioequivalence Investigation of Tadalafil-Loaded Orodispersible Films in Human Volunteers
Abstract
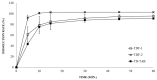
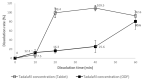
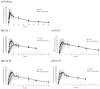
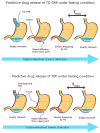
Tadalafil (TD) has poor water solubility but is well absorbed without affecting food intake when administered orally. Owing to patient adherence and therapeutic characteristics, a TD-loaded orodispersible film (TDF) is preferable. However, the mechanistic role of dietary status on the clinical pharmacokinetic analysis of TDF in human volunteers should be investigated because the gastrointestinal environment varies periodically according to meal intervals, although commercial 20 mg TD-loaded tablets (TD-TAB, Cialis® tablet) may be taken with or without food. TDF was prepared by dispersing TD in an aqueous solution and polyethylene glycol 400 to ensure good dispersibility of the TD particles. In the fasting state, each T/R of Cmax and AUC between TD-TAB and TDF showed bioequivalence with 0.936-1.105 and 1.012-1.153, respectively, and dissolution rates in 1000 mL water containing 0.5% SLS were equivalent. In contrast, TDF was not bioequivalent to TD-TAB under the fed conditions by the Cmax T/R of 0.610-0.798. The increased dissolution rate of TDF via the micronization of drug particles and the reduced viscosity of the second meal content did not significantly affect the bioequivalence. Interestingly, an increase in second meal intake time from 4 h to 6 h resulted in the bioequivalence by the Cmax T/R of 0.851-0.998 of TD-TAB and TDF. The predictive diffusion direction model for physical digestion of TD-TAB and TDF in the stomach after the first and second meal intake was successfully simulated using computational fluid dynamics modeling, accounting for the delayed drug diffusion of TDF caused by prolonged digestion of stomach contents under postprandial conditions.
Keywords: computational fluid dynamics; drug particle size; in vitro dissolution; in vivo bioequivalence; meal viscosity; orodispersible film; postprandial condition; second meal intake time; tadalafil.
Conflict of interest statement
Mr. Su-Jun Park is an employee, and received experimental costs of CTCBIO Inc. Dr. Myung-Chul Gil and Mr. Bong-Sang Lee are employees of Pluto Inc. These companies had no role in the design of the study; in the collection, analysis, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results. The remaining authors declare that they have no known competing financial interests or personal relationships that could have influenced the work reported in this study.
Figures


Similar articles
-
Development of novel tenofovir disoproxil phosphate salt with stability enhancement and bioequivalence to the commercial tenofovir disoproxil fumarate salt in rats and beagle dogs.Int J Pharm. 2020 Feb 25;576:118957. doi: 10.1016/j.ijpharm.2019.118957. Epub 2019 Dec 14. Int J Pharm. 2020. PMID: 31843551
-
Development of stabilized tenofovir disoproxil tablet: degradation profile, stabilization, and bioequivalence in beagle dogs.Drug Dev Ind Pharm. 2018 May;44(5):757-766. doi: 10.1080/03639045.2017.1411943. Epub 2017 Dec 25. Drug Dev Ind Pharm. 2018. PMID: 29198140
-
Use of acidifier and solubilizer in tadalafil solid dispersion to enhance the in vitro dissolution and oral bioavailability in rats.Int J Pharm. 2017 Jun 30;526(1-2):77-87. doi: 10.1016/j.ijpharm.2017.04.056. Epub 2017 Apr 24. Int J Pharm. 2017. PMID: 28450170
-
Design of PVP/VA S-630 based tadalafil solid dispersion to enhance the dissolution rate.Eur J Pharm Sci. 2017 Jan 15;97:269-276. doi: 10.1016/j.ejps.2016.11.030. Epub 2016 Dec 2. Eur J Pharm Sci. 2017. PMID: 27919702
-
Pharmacokinetics of a novel orodispersible tablet of sildenafil in healthy subjects.Clin Ther. 2014 Feb 1;36(2):236-44. doi: 10.1016/j.clinthera.2013.12.010. Epub 2014 Jan 18. Clin Ther. 2014. PMID: 24447534 Clinical Trial.
References
-
- Patel A.A., Patel R.J., Mishra P. Nanosuspension for Oral Delivery of Tadalafil: Pharmacodynamic and Pharmacokinetic Studies. J. Drug Deliv. Sci. Technol. 2021;61:102203. doi: 10.1016/j.jddst.2020.102203. - DOI
-
- Anderson N.R., Hartauer K.J., Kral M.A., Stephenson G.A. Beta-Carboline Drug Products. 6,821,975 B1. U.S. Patent. 2004 November 23;
-
- Patterson B., Bedding A., Jewell H., Payne C., Mitchell M. Dose-Normalised Pharmacokinetics of Tadalafil Administered as a Single Dose to Healthy Volunteers. Eur. Urol. Suppl. Off. J. Eur. Assoc. Urol. 2002;1:152. doi: 10.1016/S1569-9056(02)80593-0. - DOI
-
- von Keitz A., Rajfer J., Segal S., Murphy A., Denne J., Costigan T., Lockhart D., Beasley C.M., Emmick J.T. A Multicenter, Randomized, Double-Blind, Crossover Study to Evaluate Patient Preference between Tadalafil and Sildenafil. Eur. Urol. 2004;45:499–509. doi: 10.1016/j.eururo.2003.11.030. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

