Pirfenidone in Idiopathic Pulmonary Fibrosis: Real-World Observation on Efficacy and Safety, Focus on Patients Undergoing Antithrombotic and Anticoagulant
- PMID: 39065780
- PMCID: PMC11280355
- DOI: 10.3390/ph17070930
Pirfenidone in Idiopathic Pulmonary Fibrosis: Real-World Observation on Efficacy and Safety, Focus on Patients Undergoing Antithrombotic and Anticoagulant
Abstract
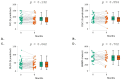
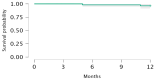
Idiopathic pulmonary fibrosis (IPF) is a rare and progressive interstitial lung disease characterized by irreversible distortion of lung architecture and subsequent loss of pulmonary function. Pirfenidone is an antifibrotic agent associated with increased progression-free survival and overall survival rates, but it carries multiple side effects. The aim of the study was to examine the efficacy and safety profile of pirfenidone in a real-life context, with a focus on the concomitant use of antithrombotic and/or anticoagulant treatments. The clinical and functional data (forced vital capacity [FVC], forced expiratory volume in 1 s [FEV1], diffusing lung capacity for carbon monoxide [DLCO], and 6 min walking test distance [6MWD]) of all IPF patients treated with pirfenidone and referred to our two centers between 2019 and 2022 were retrospectively analyzed at baseline, 6 and 12 months after the start of treatment. A total of 55 IPF subjects undergoing pirfenidone treatment were included in the analysis (45.5% females, median [IQR] age at disease onset 68.0 [10.0] years, median [IQR] age at baseline 69.0 [10.8] years). Compared to baseline, at 12 months, FVC (86.0% vs. 80.0%; p = 0.023) and DLCO (44.0% vs. 40.0%; p = 0.002) were significantly reduced, while FEV1 (p = 0.304) and 6MWD (p = 0.276) remained stable; no significant change was recorded at 6 months. Most of the reported adverse events were mild or moderate. Gastrointestinal intolerance (9.1%) was the main cause of treatment discontinuation. A total of 5% of patients reported at least one minor bleeding event, although all episodes occurred in those receiving concomitant antithrombotic or anticoagulant. Overall, this real-life experience confirms the efficacy and safety profile of pirfenidone in the case of the concomitant use of antithrombotic and/or anticoagulant drugs.
Keywords: anticoagulants; antithrombotic treatment; idiopathic pulmonary fibrosis (IPF); interstitial lung disease (ILD); pirfenidone; pulmonary function test (PFT).
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- Gahl W.A., Brantly M., Kaiser-Kupfer M.I., Iwata F., Hazelwood S., Shotelersuk V., Duffy L.F., Kuehl E.M., Troendle J., Bernardini I. Genetic defects and clinical characteristics of patients with a form of oculocutaneous albinism (Hermansky-Pudlak syndrome) N. Engl. J. Med. 1998;338:1258–1264. doi: 10.1056/NEJM199804303381803. - DOI - PubMed
LinkOut - more resources
Full Text Sources

