Relationship between Endotoxin Content in Vaccine Preclinical Formulations and Animal Welfare: An Extensive Study on Historical Data to Set an Informed Threshold
- PMID: 39066452
- PMCID: PMC11281343
- DOI: 10.3390/vaccines12070815
Relationship between Endotoxin Content in Vaccine Preclinical Formulations and Animal Welfare: An Extensive Study on Historical Data to Set an Informed Threshold
Abstract
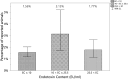
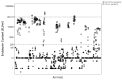
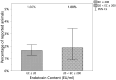
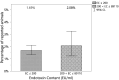
The most widely known pyrogen impurity in vaccines is the Gram-negative bacterial endotoxin lipopolysaccharide (LPS). When administered at toxic doses, endotoxin triggers inflammatory responses, which lead to endotoxic shock. The literature on endotoxic content (EC) for preclinical vaccines' formulations used in animal studies is very poor, and the recommended thresholds are solely based on commercial vaccine limits set for humans and are, therefore, not connected to the actual impact of EC on animal welfare for species used in preclinical research studies. An extensive study to evaluate the presence of a potential relationship between endotoxin content in formulations administered to mice (the most common species used in preclinical research studies) and their welfare was conducted to calculate an EC threshold for formulations of candidate vaccines. Three years of historical data, from more than 500 formulations of different antigen types (i.e., proteins, glycoconjugates, OMV/GMMA) injected into more than 5000 mice, was evaluated with two alternative statistical methodologies, both demonstrating that there is no significant relationship between actual endotoxin levels and mouse welfare. The calculation of thresholds was, therefore, performed by consistency versus formulations that demonstrated no impact on animal welfare.
Keywords: animal welfare; endotoxins; in vivo; preclinical studies; threshold.
Conflict of interest statement
This work was sponsored by GlaxoSmithKline Biologicals SA and received no external funding. The sponsor was involved in all stages of the study. All the authors are employees of the GSK group of companies. F.B., D.S. and R.C. hold company shares.
Figures

References
-
- Kiros T.G., Levast B., Auray G., Strom S., van Kessel J., Gerdts V. Innovation in Vaccinology. Spring; Berlin/Heidelberg, Germany: 2012. The Importance of Animal Models in the Development of Vaccines; pp. 251–264. - DOI
LinkOut - more resources
Full Text Sources

