Dysfunction of motor cortices in Parkinson's disease
- PMID: 39066504
- PMCID: PMC11281850
- DOI: 10.1093/cercor/bhae294
Dysfunction of motor cortices in Parkinson's disease
Abstract
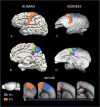
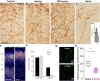
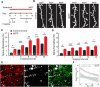
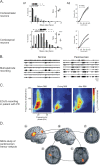
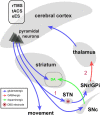
The cerebral cortex has long been thought to be involved in the pathophysiology of motor symptoms of Parkinson's disease. The impaired cortical function is believed to be a direct and immediate effect of pathologically patterned basal ganglia output, mediated to the cerebral cortex by way of the ventral motor thalamus. However, recent studies in humans with Parkinson's disease and in animal models of the disease have provided strong evidence suggesting that the involvement of the cerebral cortex is much broader than merely serving as a passive conduit for subcortical disturbances. In the present review, we discuss Parkinson's disease-related changes in frontal cortical motor regions, focusing on neuropathology, plasticity, changes in neurotransmission, and altered network interactions. We will also examine recent studies exploring the cortical circuits as potential targets for neuromodulation to treat Parkinson's disease.
Keywords: Parkinson’s disease; basal ganglia; cerebral cortex; dopamine; pathophysiology.
© The Author(s) 2024. Published by Oxford University Press.
Figures

References
-
- Andersen KB, Hansen AK, Sommerauer M, Fedorova TD, Knudsen K, Vang K, Van Den Berge N, Kinnerup M, Nahimi A, Pavese N, et al. . Altered sensorimotor cortex noradrenergic function in idiopathic REM sleep behaviour disorder - a PET study. Parkinsonism Relat Disord. 2020:75:63–69. 10.1016/j.parkreldis.2020.05.013. - DOI - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

