Improvements in hematologic markers and decreases in fatigue with pegcetacoplan for patients with paroxysmal nocturnal hemoglobinuria and mild or moderate anemia (hemoglobin ≥10 g/dL) who had received eculizumab or were naive to complement inhibitors
- PMID: 39079163
- PMCID: PMC11285951
- DOI: 10.1371/journal.pone.0306407
Improvements in hematologic markers and decreases in fatigue with pegcetacoplan for patients with paroxysmal nocturnal hemoglobinuria and mild or moderate anemia (hemoglobin ≥10 g/dL) who had received eculizumab or were naive to complement inhibitors
Abstract
Background: Although complement component 5 inhibitors (C5is) eculizumab and ravulizumab improve paroxysmal nocturnal hemoglobinuria (PNH) outcomes, patients may experience persistent anemia. This post hoc analysis investigated whether the complement component 3-targeted therapy pegcetacoplan also improved hematologic outcomes and reduced fatigue in patients with PNH and mild/moderate anemia.
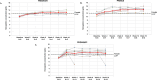
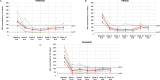
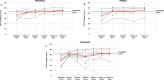
Methods: Patients with PNH and hemoglobin ≥10.0 g/dL at baseline of PADDOCK (N = 6), PRINCE (N = 8), and PEGASUS (N = 11) were included. Before receiving pegcetacoplan, PADDOCK and PRINCE patients were C5i-naive; PEGASUS patients had hemoglobin <10.5 g/dL despite stably dosed eculizumab. Hemoglobin concentrations, percentages of patients with concentrations ≥12 g/dL, and sex-specific normalization were assessed at baseline and after 16 weeks of pegcetacoplan, as were absolute reticulocyte counts (ARCs) and normalization and fatigue scores and normalization.
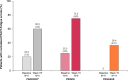
Results: From baseline to week 16, mean (SD) hemoglobin concentrations increased in C5i-naive patients (PADDOCK: 10.5 [0.4] to 12.7 [1.1] g/dL; PRINCE: 11.3 [1.0] to 14.0 [1.3] g/dL) and those with suboptimal eculizumab responses (PEGASUS: 10.2 [0.2] to 12.8 [2.6] g/dL). Percentage of patients with hemoglobin ≥12 g/dL increased (PADDOCK: 0 to 60.0% [3 of 5 patients]; PRINCE: 25.0% [2 of 8] to 87.5% [7 of 8]; PEGASUS: 0 to 72.7% [8 of 11]). Sex-specific hemoglobin normalization at week 16 occurred in 40.0% (2 of 5) (PADDOCK), 62.5% (5 of 8) (PRINCE), and 63.6% (7 of 11) (PEGASUS). In all studies, mean ARCs decreased from above normal to normal and ARC normalization increased. Mean Functional Assessment of Chronic Illness Therapy-Fatigue scores improved from below to above or near normal. Two patients had serious adverse events (PEGASUS: post-surgery sepsis, breakthrough hemolysis); breakthrough hemolysis resolved without study discontinuation.
Conclusion: Patients with PNH and mild/moderate anemia who were C5i-naive or who had suboptimal hemoglobin concentrations despite eculizumab treatment had improved hematologic outcomes and reduced fatigue after initiating or switching to pegcetacoplan.
Trial registration: Trial registration numbers: PADDOCK (NCT02588833), PRINCE (NCT04085601; EudraCT, 2018-004220-11), PEGASUS (NCT03500549).
Copyright: © 2024 Panse et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
JP: reports consultancy and honoraria at Blueprint Medicines, Amgen, F Hoffmann-La Roche, MSD, Bristol Myers Squibb, Alexion Pharmaceuticals, Novartis, Pfizer, Gilead, Boehringer Ingelheim, Swedish Orphan Biovitrum, and Apellis Pharmaceuticals; and honoraria from Swiss Biopharma. ND: has nothing to disclose SO: has nothing to disclose RPdL: has nothing to disclose PS: reports membership on an entity’s Board of Directors or advisory committees with Blueprint Medicines and Swedish Orphan Biovitrum AB; Honoraria, Membership on an entity’s Board of Directors or advisory committees and Speakers Bureau with Alexion and Bristol Myers Squibb; Honoraria and Membership on an entity’s Board of Directors or advisory committees with MSD and Novartis. NS: reports membership of advisory committee with Alexion. MA-A: was an employee at the time of this study and held stock options. EP: is an employee of Swedish Orphan Biovitrum AB. RSMW: has received consulting fees, honoraria, research funding, and speaker’s bureau fees from Alexion and F. Hoffmann-La Roche Ltd.; and research funding and speaker’s bureau fees from Apellis. These affiliations do not alter our adherence to PLOS ONE policies on sharing data and materials.
Figures




References
-
- Risitano AM, Marotta S, Ricci P, Marano L, Frieri C, Cacace F, et al.. Anti-complement treatment for paroxysmal nocturnal hemoglobinuria: time for proximal complement inhibition? A position paper from the SAAWP of the EBMT. Front Immunol. 2019;10: 1157. doi: 10.3389/fimmu.2019.01157 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

