Beneficial effects of physical exercise and an orally active mGluR2/3 antagonist pro-drug on neurogenesis and behavior in an Alzheimer's amyloidosis model
- PMID: 39081972
- PMCID: PMC11285632
- DOI: 10.3389/frdem.2023.1198006
Beneficial effects of physical exercise and an orally active mGluR2/3 antagonist pro-drug on neurogenesis and behavior in an Alzheimer's amyloidosis model
Abstract
Background: Modulation of physical activity represents an important intervention that may delay, slow, or prevent mild cognitive impairment (MCI) or dementia due to Alzheimer's disease (AD). One mechanism proposed to underlie the beneficial effect of physical exercise (PE) involves the apparent stimulation of adult hippocampal neurogenesis (AHN). BCI-838 is a pro-drug whose active metabolite BCI-632 is a negative allosteric modulator at group II metabotropic glutamate receptors (mGluR2/3). We previously demonstrated that administration of BCI-838 to a mouse model of brain accumulation of oligomeric AβE22Q (APP E693Q = "Dutch APP") reduced learning behavior impairment and anxiety, both of which are associated with the phenotype of Dutch APP mice.
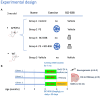
Methods: 3-month-old mice were administered BCI-838 and/or physical exercise for 1 month and then tested in novel object recognition, neurogenesis, and RNAseq.
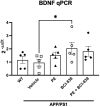
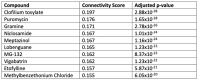
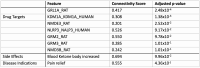
Results: Here we show that (i) administration of BCI-838 and a combination of BCI-838 and PE enhanced AHN in a 4-month old mouse model of AD amyloid pathology (APP KM670/671NL /PSEN1 Δexon9= APP/PS1), (ii) administration of BCI-838 alone or with PE led to stimulation of AHN and improvement in recognition memory, (iii) the hippocampal dentate gyrus transcriptome of APP/PS1 mice following BCI-838 treatment showed up-regulation of brain-derived neurotrophic factor (BDNF), PIK3C2A of the PI3K-mTOR pathway, and metabotropic glutamate receptors, and down-regulation of EIF5A involved in modulation of mTOR activity by ketamine, and (iv) validation by qPCR of an association between increased BDNF levels and BCI-838 treatment.
Conclusion: Our study points to BCI-838 as a safe and orally active compound capable of mimicking the beneficial effect of PE on AHN and recognition memory in a mouse model of AD amyloid pathology.
Keywords: AD amyloid pathology; APP/PS1 mice; BDNF; mGlu2/3 antagonist; neurogenesis; physical exercise.
Copyright © 2023 Perez Garcia, Bicak, Buros, Haure-Mirande, Perez, Otero-Pagan, Gama Sosa, De Gasperi, Sano, Gage, Barlow, Dudley, Glicksberg, Wang, Readhead, Ehrlich, Elder and Gandy.
Conflict of interest statement
JB was employed by Generable, Inc. CB was employed by E-Scape Bio. MS has served as a consultant for Bayer Schering Pharma, Bristol-Meyers Squibb, Elan Corporation, Genentech, Medivation, Medpace, Pfizer, Janssen, Takeda Pharmaceutical Company Limited, and United Biosource Corporation. She receives research support from the NIH. FHG was a founder and member of the SAB for BCI, but he no longer serves as a consultant or has stock because BCI no longer exists as a company. He has received funding from NIA, HIH, and NIMH on projects related to adult neurogenesis but not related to this or related compounds. CB is a former employee of BrainCells, Inc. BrainCells, Inc. provided drug and advice. JD has served as a consultant for Janssen and is an equity holder in Thorne HealthTech. BG is a consultant for Anthem AI and a scientific advisor and consultant for Prometheus Biosciences. He has received consulting fees from GLG Research and honoraria from Virtual EP Connect. ME receives research support from the NIH, the XDP Foundation, and the Cure Alzheimer's Fund. SG is a co-founder of Recuerdo Pharmaceuticals. He has served as a consultant in the past for J&J, Diagenic, and Pfizer, and he currently consults for Cognito Therapeutics, GLG Group, SVB Securities, Guidepoint, Third Bridge, Leerink, MEDACORP, Altpep, Vigil Neurosciences, and Eisai. He has received research support from Warner-Lambert, Pfizer, Baxter Healthcare, Amicus, Avid, and ADDF. He served on the DSMB for an amyloid vaccine trial by Elan Pharmaceuticals. He receives research support from the VA, NIH, and the Cure Alzheimer's Fund. SG and ME have received compensation for chart review in the areas of cognitive neurology and pediatric neurology, respectively. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) SG, BG, FHG, MG, and J-VH-M declare that they were editors board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures




References
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

