Making sense of scattering: Seeing microstructure through shear waves
- PMID: 39083612
- PMCID: PMC11290519
- DOI: 10.1126/sciadv.adp3363
Making sense of scattering: Seeing microstructure through shear waves
Abstract
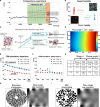
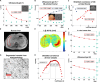
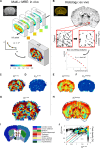
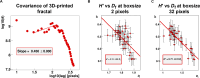
The physics of shear waves traveling through matter carries fundamental insights into its structure, for instance, quantifying stiffness for disease characterization. However, the origin of shear wave attenuation in tissue is currently not properly understood. Attenuation is caused by two phenomena: absorption due to energy dissipation and scattering on structures such as vessels fundamentally tied to the material's microstructure. Here, we present a scattering theory in conjunction with magnetic resonance imaging, which enables the unraveling of a material's innate constitutive and scattering characteristics. By overcoming a three-order-of-magnitude scale difference between wavelength and average intervessel distance, we provide noninvasively a macroscopic measure of vascular architecture. The validity of the theory is demonstrated through simulations, phantoms, in vivo mice, and human experiments and compared against histology as gold standard. Our approach expands the field of imaging by using the dispersion properties of shear waves as macroscopic observable proxies for deciphering the underlying ultrastructures.
Figures
References
-
- Schmitt J. M., Kumar G., Turbulent nature of refractive-index variations in biological tissue. Opt. Lett. 21, 1310–1312 (1996). - PubMed
-
- Xu M., Alfano R. R., Fractal mechanisms of light scattering in biological tissue and cells. Opt. Lett. 30, 3051–3053 (2005). - PubMed
-
- Barthelemy P., Bertolotti J., Wiersma D. S., A Lévy flight for light. Nature 453, 495–498 (2008). - PubMed
-
- Page J. H., Sheng P., Schriemer H. P., Jones I., Jing X., Weitz D. A., Group velocity in strongly scattering media. Science 271, 634–637 (1996).
-
- Tourin A., Fink M., Derode A., Multiple scattering of sound. Waves Random Media 10, R31 (2000).
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

