In-vitro safety assessment of meropenem on human retinal pigment epithelium (RPE)
- PMID: 39092242
- PMCID: PMC11292269
- DOI: 10.1016/j.heliyon.2024.e33916
In-vitro safety assessment of meropenem on human retinal pigment epithelium (RPE)
Abstract
Purpose: Endophthalmitis is a severe infection accompanied by inflammation that affects the anterior and posterior parts of the eye. It is typically treated with a combination of antibiotics that cover various microorganisms. However, retinal pigment epithelium (RPE) cells are highly susceptible to damage from intravitreal injection therapy. This study aimed to investigate the impact of clinically relevant concentrations of meropenem (alone or in combination with vancomycin) on the viability and inflammation of RPE cells.
Design: In-vitro Study.
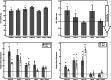
Methods: RPE cells from passages 5-7 were treated with different concentrations of meropenem (1/4x, x, and 4x; [x = 16 mg/L]), vancomycin (30 mg/L), and meropenem (x) plus vancomycin for 24 h. The morphology assessment and MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay were performed to evaluate cytotoxicity due to drug treatment. Real-time PCR was used to measure the relative expression of apoptotic genes (BCL-2 and BAX) and inflammation biomarkers (IL-1b and IL-6).
Results: Meropenem (alone or in combination with vancomycin) did not have any significant effect on RPE cell morphology, density, and viability. Gene expression analysis confirmed these results, showing no significant changes in the BCL-2/BAX ratio in drug-treated RPE cells compared to controls. Treatment with meropenem significantly induced the expression of IL-1b at all studied concentrations. Additionally, at concentrations of x and 4x, it also significantly increased the expression of IL-6, which was dose-dependent. However, this effect was not observed with vancomycin alone or in combination with meropenem.
Conclusions: The results of this study suggest that meropenem, either alone or in combination with vancomycin, does not induce RPE cytotoxicity. There was an upregulation of IL-1b and IL-6 in meropenem monotherapy, the clinical implication of which should be elucidated in future in-vivo or clinical studies.
Keywords: Apoptosis; Endophthalmitis; Inflammation; Meropenem; RPE.
© 2024 The Authors.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures


References
-
- Sansome S.G., Ting M., Jain S. Endophthalmitis. Br. J. Hosp. Med. 2019;80(1):C8–C11. - PubMed
-
- Ferreira F.B.C. 2023. Endophthalmitis: a Threat to the Eye.
LinkOut - more resources
Full Text Sources
Research Materials

