11β-Hydroxysteroid dehydrogenase type 2 may mediate the stress-specific effects of cortisol on brain cell proliferation in adult zebrafish (Danio rerio)
- PMID: 39092490
- PMCID: PMC11418181
- DOI: 10.1242/jeb.248020
11β-Hydroxysteroid dehydrogenase type 2 may mediate the stress-specific effects of cortisol on brain cell proliferation in adult zebrafish (Danio rerio)
Abstract
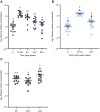
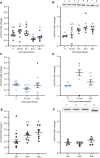
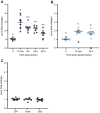
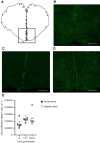
Stress-induced increases in cortisol can stimulate or inhibit brain cell proliferation, but the mechanisms behind these opposing effects are unknown. We tested the hypothesis that 11β-hydroxysteroid dehydrogenase type 2 (Hsd11b2), a glucocorticoid-inactivating enzyme expressed in neurogenic regions of the adult zebrafish brain, mitigates cortisol-induced changes to brain cell proliferation, using one of three stress regimes: a single 1 min air exposure (acute stress), two air exposures spaced 24 h apart (repeat acute stress) or social subordination (chronic stress). Plasma cortisol was significantly elevated 15 min after air exposure and recovered within 24 h after acute and repeat acute stress, whereas subordinate fish exhibited significant and sustained elevations relative to dominant fish for 24 h. Following acute stress, brain hsd11b2 transcript abundance was elevated up to 6 h after a single air exposure but was unchanged by repeat acute stress or social subordination. A sustained increase in brain Hsd11b2 protein levels occurred after acute stress, but not after repeat or chronic stress. Following acute and repeat acute stress, brain pcna transcript abundance (a marker of cell proliferation) exhibited a prolonged elevation, but was unaffected by social subordination. Interestingly, the number of telencephalic BrdU+ cells increased in fish after a single air exposure but was unchanged by repeat acute stress. Following acute and repeat acute stress, fish expressed lower brain glucocorticoid and mineralocorticoid receptor (gr and mr) transcript abundance while subordinate fish exhibited no changes. Taken together, these results demonstrate stressor-specific regulation of Hsd11b2 in the zebrafish brain that could modulate rates of cortisol catabolism contributing to observed differences in brain cell proliferation.
Keywords: Adult neurogenesis; Glucocorticoid regulation; Neuroendocrinology; Neuroplasticity; Regulation of mitosis; Stress biology.
© 2024. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interests The authors declare no competing or financial interests.
Figures


References
-
- Ausas, M. S., Mazzitelli-Fuentes, L., Román, F. R., Crichigno, S. A., De Vincenti, A. P. and Mongiat, L. A. (2019). Social isolation impairs active avoidance performance and decreases neurogenesis in the dorsomedial telencephalon of rainbow trout. Physiol. Behav. 198, 1-10. 10.1016/j.physbeh.2018.10.006 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

