O-glycosylation of IgA1 and the pathogenesis of an autoimmune disease IgA nephropathy
- PMID: 39095059
- PMCID: PMC11442006
- DOI: 10.1093/glycob/cwae060
O-glycosylation of IgA1 and the pathogenesis of an autoimmune disease IgA nephropathy
Abstract
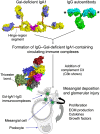
IgA nephropathy is a kidney disease characterized by deposition of immune complexes containing abnormally O-glycosylated IgA1 in the glomeruli. Specifically, some O-glycans are missing galactose that is normally β1,3-linked to N-acetylgalactosamine of the core 1 glycans. These galactose-deficient IgA1 glycoforms are produced by IgA1-secreting cells due to a dysregulated expression and activity of several glycosyltransferases. Galactose-deficient IgA1 in the circulation of patients with IgA nephropathy is bound by IgG autoantibodies and the resultant immune complexes can contain additional proteins, such as complement C3. These complexes, if not removed from the circulation, can enter the glomerular mesangium, activate the resident mesangial cells, and induce glomerular injury. In this review, we briefly summarize clinical and pathological features of IgA nephropathy, review normal and aberrant IgA1 O-glycosylation pathways, and discuss the origins and potential significance of natural anti-glycan antibodies, namely those recognizing N-acetylgalactosamine. We also discuss the features of autoantibodies specific for galactose-deficient IgA1 and the characteristics of pathogenic immune complexes containing IgA1 and IgG. In IgA nephropathy, kidneys are injured by IgA1-containing immune complexes as innocent bystanders. Most patients with IgA nephropathy progress to kidney failure and require dialysis or transplantation. Moreover, most patients after transplantation experience a recurrent disease. Thus, a better understanding of the pathogenetic mechanisms is needed to develop new disease-specific treatments.
Keywords: IgA nephropathy; IgA1 O-glycans; autoimmune disease; immune complexes.
© The Author(s) 2024. Published by Oxford University Press. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures


References
-
- Abeygunawardana C, Bush CA, Cisar JO. Complete structure of the cell surface polysaccharide of Streptococcus oralis C104: a 600-MHz NMR study. Biochemistry. 1991:30(35):8568–8577. - PubMed
-
- Allen AC. Abnormal glycosylation of IgA: is it related to the pathogenesis of IgA nephropathy? Nephrol Dial Transplant. 1995:10(7):1121–1124. - PubMed
-
- Allen AC. Methodological approaches to the analysis of IgA1 O-glycosylation in IgA nephropathy. J Nephrol. 1999:12(2):76–84. - PubMed
-
- Allen AC, Bailey EM, Brenchley PEC, Buck KS, Barratt J, Feehally J. Mesangial IgA1 in IgA nephropathy exhibits aberrant O-glycosylation: observations in three patients. Kidney Int. 2001:60(3):969–973. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

