Electrophysiological Responses of Trissolcus japonicus, T. basalis, and T. oenone (Hymenoptera: Scelionidae) to Volatile Compounds Associated with New Zealand Stink Bugs (Hemiptera: Pentatomidae)
- PMID: 39095554
- PMCID: PMC11717797
- DOI: 10.1007/s10886-024-01533-7
Electrophysiological Responses of Trissolcus japonicus, T. basalis, and T. oenone (Hymenoptera: Scelionidae) to Volatile Compounds Associated with New Zealand Stink Bugs (Hemiptera: Pentatomidae)
Abstract
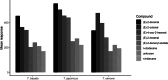
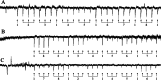
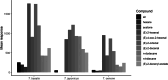
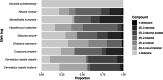
Parasitoid biological control agents rely heavily on olfaction to locate their hosts. Chemical cues associated with hosts and non-hosts are known to influence the expression of host preferences and host-specificity. A better understanding of how and why parasitoids attack some species and not others, based on volatile organic compounds associated with potential hosts, can provide key information on the parasitoid's host preferences, which could be applied to pre-release risk assessments for classical biological control agents. Electrophysiological techniques such as electroantennography (EAG) and GC-EAD (gas chromatography coupled with electroantennographic detection) are widely used to identify bioactive semiochemicals. But the application of these techniques to understanding how chemical ecological cues mediate parasitoid host specificity has not been as thoroughly explored. We conducted GC-EAD and EAG studies to identify olfactory-active compounds associated with adult females of nine stink bug species from Aotearoa/New Zealand on the antennae of three closely related parasitoid species: Trissolcus japonicus Ashmead, a pre-emptively (= proactively) approved biocontrol agent against brown marmorated stink bug; T. basalis (Wollaston), a biocontrol agent introduced against Nezara viridula L. in 1949; and T. oenone Johnson, a native Australasian pentatomid parasitoid. Eight compounds associated with stink bugs elicited antennal responses from all three parasitoids, and we were able to identify seven of these. (E)-2-hexenal, (E)-4-oxo-2-hexenal, (E)-2-octenal and (E)-2-decenal generally elicited stronger responses in the three parasitoids, while n-tridecane, n-dodecane, and (E)-2-decenyl acetate elicited weaker responses. We discuss how and why the results from electrophysiological experiments can be applied to non-target risk assessments within biological control programmes.
Keywords: Electrophysiology; Host Chemistry; Host Range; Non-Target Risks; Parasitism.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Competing Interests: The authors declare no competing interests.
Figures



Similar articles
-
Integrating Chemical Ecology with Behavioural Bioassays to Understand Host Preferences in the Stink Bug Egg Parasitoids Trissolcus basalis and Trissolcus oenone (Hymenoptera: Scelionidae).J Chem Ecol. 2025 May 28;51(3):60. doi: 10.1007/s10886-025-01616-z. J Chem Ecol. 2025. PMID: 40425981 Free PMC article.
-
Response of the egg parasitoids Trissolcus basalis and Telenomus podisi to compounds from defensive secretions of stink bugs.J Chem Ecol. 2009 Jan;35(1):8-19. doi: 10.1007/s10886-008-9578-0. Epub 2009 Jan 7. J Chem Ecol. 2009. PMID: 19127384
-
Is It Possible to Manipulate Scelionidae Wasps' Preference to a Target Host?Neotrop Entomol. 2018 Oct;47(5):689-697. doi: 10.1007/s13744-018-0607-6. Epub 2018 Apr 20. Neotrop Entomol. 2018. PMID: 29679311
-
What's my age again? Assessing the impact of stink bug egg mass age on host recognition by egg parasitoids Trissolcus basalis and Trissolcus japonicus (Hymenoptera: Scelionidae).Front Physiol. 2025 Jul 17;16:1588946. doi: 10.3389/fphys.2025.1588946. eCollection 2025. Front Physiol. 2025. PMID: 40746985 Free PMC article.
-
A Comprehensive Survey of Stink Bug Pheromones - Extraction, Identification, Synthesis, Biosynthesis, and Phylogenetic Insights.Chem Rec. 2025 Apr;25(4):e202400140. doi: 10.1002/tcr.202400140. Epub 2024 Nov 19. Chem Rec. 2025. PMID: 39562170 Review.
Cited by
-
Integrating Chemical Ecology with Behavioural Bioassays to Understand Host Preferences in the Stink Bug Egg Parasitoids Trissolcus basalis and Trissolcus oenone (Hymenoptera: Scelionidae).J Chem Ecol. 2025 May 28;51(3):60. doi: 10.1007/s10886-025-01616-z. J Chem Ecol. 2025. PMID: 40425981 Free PMC article.
References
-
- Aldrich JR (1988) Chemical ecology of the Heteroptera. Ann Rev Entomol 33:211–238. 10.1146/annurev.en.33.010188.001235
-
- Aldrich JR (1995) Chemical communication in the true bugs and parasitoid exploitation. In: Cardé RT, Bell WJ (eds) Chemical Ecology of Insects 2. Springer, US, Boston, MA, pp 318–363
-
- Aldrich JR, Oliver JE, Waite GK et al (1996) Identification of presumed pheromone blend from Australasian predaceous bug, Oechalia schellenbergii (Heteroptera: Pentatomidae). J Chem Ecol 22:729–738. 10.1007/BF02033582 - PubMed
-
- Arn H, Städler E, Rauscher S (1975) The electroantennographic detector — a selective and sensitive tool in the gas chromatographic analysis of insect pheromones. Z Naturforsch C 30:722–725. 10.1515/znc-1975-11-1204
-
- Austin AD, Johnson NF, Dowton M (2005) Systematics, evolution, and biology of scelionid and platygastrid wasps. Ann Rev Entomol 50:553–582. 10.1146/annurev.ento.50.071803.130500 - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

