Clinical, Genomic, and Transcriptomic Characteristics of Patients with Metastatic Renal Cell Carcinoma Who Developed Thromboembolic Events
- PMID: 39100549
- PMCID: PMC11296887
- DOI: 10.15586/jkcvhl.v11i3.319
Clinical, Genomic, and Transcriptomic Characteristics of Patients with Metastatic Renal Cell Carcinoma Who Developed Thromboembolic Events
Abstract
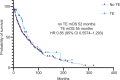
Thromboembolic events (TE) are a common complication in patients with metastatic renal cell carcinoma (mRCC) and are associated with poorer clinical outcomes. However, the incidence of TE and clinical and genomic characteristics of patients with mRCC who develop this complication are poorly understood. Herein, we describe the incidence and clinical features of patients with mRCC with or without TE at our institution, and examine their association with the underlying genomic and transcriptomic characteristics of the tumor. This retrospective study included all consecutive cases of mRCC seen at our institution. A CLIA-certified lab performed tumor genomics and transcriptomics. Patients were classified based on the presence of a TE within the first year of diagnosis. Three hundred and seventy patients with mRCC were included in the study. TE was seen in 11% (42) of the patients. Patients with favorable International mRCC Database Consortium (IMDC) risk were less likely to develop a TE. In contrast, patients receiving combination treatment with a tyrosine kinase inhibitor (TKI) and an immune checkpoint inhibitor were more likely to develop a TE. No difference in overall survival among patients with or without TE was observed (52 vs. 55 months; HR 0.85, 95% CI 0.5574-1.293, p = 0.24). The most upregulated pathways in mRCC with TEs versus those without were the xenobiotic metabolism and mTORC1 signaling pathways. Our findings suggest potential biomarkers that, after external validation, could be used to better select patients who would benefit from prophylactic anticoagulation.
Keywords: cancer; kidney; renal cell carcinoma; thromboembolic events.
Copyright: Fortuna G.G., et al.
Conflict of interest statement
Neeraj Agarwal (lifetime disclosures): No personal COIs since April 15, 2021. Consultancy to Astellas, Astra Zeneca, Aveo, Bayer, Bristol Myers Squibb, Calithera, Clovis, Eisai, Eli Lilly, EMD Serono, Exelixis, Foundation Medicine, Genentech, Gilead, Janssen, Merck, MEI Pharma, Nektar, Novartis, Pfizer, Pharmacyclics, and Seattle Genetics. Research funding to Neeraj Agarwal’s institution (lifetime): Arnivas, Astellas, Astra Zeneca, Bavarian Nordic, Bayer, Bristol Myers Squibb, Calithera, Celldex, Clovis, Crispr, Eisai, Eli Lilly, EMD Serono, Exelixis, Genentech, Gilead, Glaxo Smith Kline, Immunomedics, Janssen, Lava, Medivation, Merck, Nektar, Neoleukin, New Link Genetics, Novartis, Oric, Pfizer, Prometheus, Rexahn, Roche, Sanofi, Seattle Genetics, Takeda, and Tracon. Umang Swami reports consultancy to Astellas, Exelixis, Seattle Genetics, Imvax, Sanofi, Pfizer, AstraZeneca, and Gilead and research funding to institute from Janssen, Exelixis, and Astellas/Seattle Genetics. Benjamin L. Maughan received financial compensation as a paid consultant/advisor to Abbive, Pfizer, AVEO oncology, Janssen, Astellas, Bristol-Myers Squibb, Clovis, Tempus, Merck, Exelixis, Bayer Oncology, Lilly, Sanofi, Telix, and Peloton Therapeutics; Huntsman Cancer Institute has received research funding from Exelixis, Bavarian-Nordic, Clovis, and Bristol-Myers Squibb on behalf of Benjamin L. Maughan. The rest of the authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures
References
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous