Glycogen Phosphorylase Inhibitor Promotes Hair Growth via Protecting from Oxidative-Stress and Regulating Glycogen Breakdown in Human Hair follicles
- PMID: 39103246
- PMCID: PMC11392663
- DOI: 10.4062/biomolther.2024.098
Glycogen Phosphorylase Inhibitor Promotes Hair Growth via Protecting from Oxidative-Stress and Regulating Glycogen Breakdown in Human Hair follicles
Abstract
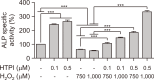
Hair growth cycles are mainly regulated by human dermal papilla cells (hDPCs) and human outer root sheath cells (hORSCs). Protecting hDPCs from excessive oxidative stress and hORSCs from glycogen phosphorylase (PYGL) is crucial to maintaining the hair growth phase, anagen. In this study, we developed a new PYGL inhibitor, Hydroxytrimethylpyridinyl Methylindolecarboxamide (HTPI) and assessed its potential to prevent hair loss. HTPI reduced oxidative damage, preventing cell death and restored decreased level of anagen marker ALP and its related genes induced by hydrogen peroxide in hDPCs. Moreover, HTPI inhibited glycogen degradation and induced cell survival under glucose starvation in hORSCs. In ex-vivo culture, HTPI significantly enhanced hair growth compared to the control with minoxidil showing comparable results. Overall, these findings suggest that HTPI has significant potential as a therapeutic agent for the prevention and treatment of hair loss.
Keywords: Anagen; Dermal papillia cells; Glycogen phosphorylase; Hair loss; Outer root sheath cells; Oxidative stress.
Figures

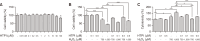
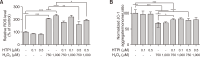


References
LinkOut - more resources
Full Text Sources
Miscellaneous

