This is a preprint.
Chromosome X-Wide Common Variant Association Study (XWAS) in Autism Spectrum Disorder
- PMID: 39108515
- PMCID: PMC11302709
- DOI: 10.1101/2024.07.18.24310640
Chromosome X-Wide Common Variant Association Study (XWAS) in Autism Spectrum Disorder
Update in
-
Chromosome X-wide common variant association study in autism spectrum disorder.Am J Hum Genet. 2025 Jan 2;112(1):135-153. doi: 10.1016/j.ajhg.2024.11.008. Epub 2024 Dec 19. Am J Hum Genet. 2025. PMID: 39706197 Free PMC article.
Abstract
Autism Spectrum Disorder (ASD) displays a notable male bias in prevalence. Research into rare (<0.1) genetic variants on the X chromosome has implicated over 20 genes in ASD pathogenesis, such as MECP2, DDX3X, and DMD. The "female protective effect" in ASD suggests that females may require a higher genetic burden to manifest similar symptoms as males, yet the mechanisms remain unclear. Despite technological advances in genomics, the complexity of the biological nature of sex chromosomes leave them underrepresented in genome-wide studies. Here, we conducted an X chromosome-wide association study (XWAS) using whole-genome sequencing data from 6,873 individuals with ASD (82% males) across Autism Speaks MSSNG, Simons Simplex Cohort SSC, and Simons Foundation Powering Autism Research SPARK, alongside 8,981 population controls (43% males). We analyzed 418,652 X-chromosome variants, identifying 59 associated with ASD (p-values 7.9×10-6 to 1.51×10-5), surpassing Bonferroni-corrected thresholds. Key findings include significant regions on chrXp22.2 (lead SNP=rs12687599, p=3.57×10-7) harboring ASB9/ASB11, and another encompassing DDX53/PTCHD1-AS long non-coding RNA (lead SNP=rs5926125, p=9.47×10-6). When mapping genes within 10kb of the 59 most significantly associated SNPs, 91 genes were found, 17 of which yielded association with ASD (GRPR, AP1S2, DDX53, HDAC8, PCDH19, PTCHD1, PCDH11X, PTCHD1-AS, DMD, SYAP1, CNKSR2, GLRA2, OFD1, CDKL5, GPRASP2, NXF5, SH3KBP1). FGF13 emerged as a novel X-linked ASD candidate gene, highlighted by sex-specific differences in minor allele frequencies. These results reveal significant new insights into X chromosome biology in ASD, confirming and nominating genes and pathways for further investigation.
Conflict of interest statement
At the time of this study and its publication, S.W.S. served on the Scientific Advisory Committee of Population Bio. Intellectual property from aspects of his research held at The Hospital for Sick Children are licensed to Athena Diagnostics and Population Bio. These relationships did not influence data interpretation or presentation during this study but are disclosed for potential future considerations.
Figures
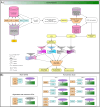
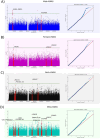




References
-
- Maenner M.J., Warren Z., Williams A.R., Amoakohene E., Bakian A.V., Bilder D.A., Durkin M.S., Fitzgerald R.T., Furnier S.M., Hughes M.M., et al. (2023). Prevalence and Characteristics of Autism Spectrum Disorder Among Children Aged 8 Years - Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2020. MMWR Surveill. Summ. 72,. 10.15585/mmwr.ss7202a1 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
