Interference of lupus anticoagulant causing antiprothrombin and anti-beta-2-glycoprotein I antibodies on international normalized ratio measurements: comparative analysis of international normalized ratio methods
- PMID: 39109234
- PMCID: PMC11301202
- DOI: 10.1016/j.rpth.2024.102470
Interference of lupus anticoagulant causing antiprothrombin and anti-beta-2-glycoprotein I antibodies on international normalized ratio measurements: comparative analysis of international normalized ratio methods
Abstract
Background: Life-long vitamin K antagonist (VKA) therapy is recommended as a standard of care in antiphospholipid syndrome (APS) patients with thrombosis. Concerns have been raised about the validity of international normalized ratio (INR) measurements in lupus anticoagulant (LA)-positive APS patients because LA may interfere with phospholipid-dependent coagulation tests and could elevate INR measurements.
Objectives: Here, we aimed to determine the interference of antigen-specific monoclonal and isolated patient antibodies with LA activity on INR measurements.
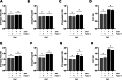
Methods: Pooled normal plasma and control plasma from patients on VKA (without LA) were incubated with monoclonal and isolated patient immunoglobulin G antiprothrombin and anti-beta-2-glycoprotein I antibodies that express LA activity. INR was determined before and after addition using 3 laboratory assays (Owren STA-Hepato Prest, Quick STA-NeoPTimal, and Quick STA-Neoplastine R) and 1 point-of-care test device (CoaguChek Pro II).
Results: Antiprothrombin and anti-beta-2-glycoprotein I antibodies with LA activity interfered with recombinant human thromboplastin reagents (Quick STA-Neoplastine R and CoaguChek Pro II), particularly when added to plasma of VKA-treated controls. This effect was most evident on point-of-care test INR measurements, while the recombinant Quick reagent exhibited a lesser degree of interference. In contrast, tissue-derived thromboplastin reagents (Owren STA-Hepato Prest and Quick STA-NeoPTimal) remained largely unaffected by these antibodies, both in pooled normal plasma and VKA anticoagulated control plasma. Among these reagents, the Owren INR reagent exhibited the lowest sensitivity to the influence of LA antibodies. This observed difference in sensitivity is independent of the plasma dilution factor or the presence of factor V or fibrinogen in Owren reagent.
Conclusion: INR reagents that utilize recombinant human thromboplastin are more sensitive to the presence of monoclonal and patient-derived antibodies with LA activity. Consequently, APS patients positive for LA should be monitored using tissue-derived thromboplastin reagents, given its reduced susceptibility to interference by LA-causing antibodies.
Keywords: anticoagulants; antiphospholipid syndrome; international normalized ratio; lupus coagulation inhibitor; warfarin.
© 2024 The Author(s).
Figures





References
-
- Miyakis S., Lockshin M.D., Atsumi T., Branch D.W., Brey R.L., Cervera R., et al. International consensus statement on an update of the classification criteria for definite antiphospholipid syndrome (APS) J Thromb Haemost. 2006;4:295–306. - PubMed
-
- Molhoek J.E., de Groot P.G., Urbanus R.T. The lupus anticoagulant paradox. Semin Thromb Hemost. 2018;44:445–452. - PubMed
-
- Della Valle P., Crippa L., Garlando A.M., Pattarini E., Safa O., D'angelo S.V., et al. Interference of lupus anticoagulants in prothrombin time assays: implications for selection of adequate methods to optimize the management of thrombosis in the antiphospholipid-antibody syndrome. Haematologica. 1999;84:1065–1074. - PubMed
-
- Oostin J.D., Derksen R.H., Entjes H.T.I., Bouma B.N., de Groot P.G. Lupus anticoagulant activity is frequently dependent on the presence of β2-glycoprotein I. Thromb Haemost. 1992;67:499–502. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

