Momelotinib versus ruxolitinib in JAK inhibitor-naïve patients with myelofibrosis: an efficacy/safety analysis in the Japanese subgroup of the phase 3 randomized SIMPLIFY-1 trial
- PMID: 39110143
- PMCID: PMC11362197
- DOI: 10.1007/s12185-024-03822-z
Momelotinib versus ruxolitinib in JAK inhibitor-naïve patients with myelofibrosis: an efficacy/safety analysis in the Japanese subgroup of the phase 3 randomized SIMPLIFY-1 trial
Abstract
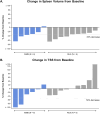
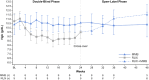
Momelotinib, an oral Janus kinase (JAK) 1/2 and activin A receptor type 1 inhibitor, improved symptoms, splenomegaly, and anemia in patients with myelofibrosis (MF). This sub-analysis of SIMPLIFY-1 evaluated the efficacy and safety of momelotinib versus ruxolitinib in Japanese patients with JAK inhibitor (JAKi)-naïve MF. Patients were randomized 1:1 to receive momelotinib 200 mg once daily or ruxolitinib 20 mg twice daily (or modified based on label) for 24 weeks, after which patients could receive open-label momelotinib. The primary endpoint was splenic response rate (SRR; ≥ 35% reduction in spleen volume) at 24 weeks; main secondary endpoints were total symptom score (TSS) response (≥ 50% reduction) and transfusion independence (TI) rates. Fifteen Japanese patients (momelotinib, n = 6; ruxolitinib, n = 9) were enrolled; all completed treatment. At Week 24, SRR was 50.0% with momelotinib and 44.4% with ruxolitinib. TSS response rates were 33.3% and 0%, and TI rates were 83.3% and 44.4%. Any-grade treatment-related adverse event (TRAE) rates were 83.3% with momelotinib and 88.9% with ruxolitinib. Grade 3/4 TRAE rates were 0% and 55.6%, with specific events being anemia (55.6%) and vertigo (11.1%) with ruxolitinib. Momelotinib was well tolerated, improved spleen and symptom responses, and reduced transfusion requirements in Japanese patients with JAKi-naïve MF.
Keywords: Japan; Momelotinib; Myelofibrosis; Phase 3; Ruxolitinib.
© 2024. The Author(s).
Conflict of interest statement
Kazuya Shimoda has employment/leadership position/advisory role with AbbVie, PharmaEssentia, Sumitomo Pharma, GSK, a honoraria role with Novartis and Takeda, received research funds AstraZeneca, PharmaEssentia, Meiji Pharma, and received subsidies/donations from Chugai Pharma, Kyowa Kirin, AbbVie, Daiichi Sankyo, Otsuka, Eisai, Shionogi and Asahi Kasei Medical. Norio Komatsu has employment/leadership position/advisory role with PharmaEssentia Japan; received fees from Takeda Pharmaceutical Co., Ltd., Novartis Pharmaceutical, Japan Tobacco Inc.; received researched funds from Takeda Pharmaceutical Co., Ltd., PerseusProteomics Inc., Meiji Seika Pharmaceutical Co., Ltd., PharmaEssentia Japan; received donated fund laboratory from Otsuka Pharmaceutical Co., Ltd., Kyowa Kirin Co., Ltd., Sumitomo Pharmaceutical Co., Ltd., PharmaEssentia Japan, Chugai Pharmaceutical Co., Ltd., Takeda Pharmaceutical Co., Ltd. Itaru Matsumura received fees from Novartis Pharmaceutical KK, Bristol-Myers Squibb Co., Ltd., Pfizer Japan Inc., Daiichi Sankyo Co., Ltd., Otsuka Pharmaceutical Co., Ltd., Astellas Pharmaceutical Inc., Janssen Pharmaceutical KK, AbbVie GK., Takeda Pharmaceutical Co., Ltd., Onopharmaceutical Co., Ltd., SymBio Pharmaceuticals Ltd.; received research funds from Kyowa Kirin Co., Ltd., Chugai Pharmaceutical Co., Ltd., Novartis Pharmaceutical KK, Astellas Pharmaceutical Inc., AbbVie GK., Pfizer Japan Inc., Takeda Pharmaceutical Co., Ltd., Alexion, Otsuka Pharmaceutical Co., Ltd., Janssen Pharmaceutical KK; received subsidies/donations from Chugai Pharmaceutical Co., Ltd., Kyowa Kirin Co., Ltd, Sumitomo Dainippon Pharmaceutical Co., Ltd., Takeda Pharmaceutical Co., Ltd., Shionogi &Co., Ltd.; Asahi Kasei Pharmaceutical Co., Eisai Co., Ltd., Taiho Pharmaceutical Co., Ltd., Nippon Shinyaku Co., Ltd., Otsuka Pharmaceutical Co., Ltd., AbbVie GK., Onopharmaceutical Co., Ltd., Sanofi KK., Mitsubishi Tanabe Pharmaceutical Corporation. Kazuhiko Ikeda received fees from Novartis, Kyokuto, PharmaEssentia Japan, Japan Blood Product Organization, Nippon Shinyaku, Otsuka, Ortho Clinical Diagnostics, Zeria, Takeda, Ono; received research funds from Kyowa-Kirin, Daiichi Sankyo, Kyokuto, Hokuyo-Denki; received reagents from GenDx. Masayuki Hino received fees from Novartis, Bristol Myers Squib, Otsuka, Kyowa-Kirin; received research funds from Labcorp Drug Development; received subsidies/donations from Chugai, Kyowa-Kirin, Otsuka, Takeda, Taiho, Sekisui Medical. Michihiro Hidaka has no COIs to declare. Yoshinobu Maeda received fees from Kyowa Kirin Co., Ltd., Novartis Pharmaceutical Co., Ltd.; received research funds from Nippon Shinyaku Co., Ltd., Chugai Pharmaceutical Co. Ltd; received subsidies/donations from Otsuka Pharmaceutical Co., Ltd., Chugai Pharmaceutical Co. Ltd., Eisai Co., Ltd., Takeda Pharmaceutical Co., Ltd., Kyowa Kirin Co. Ltd. Takeshi Kondo received fees from Pfizer, Otsuka Pharmaceuticals Co., Ltd., Novartis, Astellas. Tomoaki Fujisaki has no COIs to declare. Keita Shoshi has employment/leadership position/advisory role with GSK KK. Kyoichi Azuma has employment/leadership position/advisory role with GSK KK. Ryuichi Fukushima has employment/leadership position/advisory role with GSK KK. Jun Kawashima was employed in a leadership position/advisory role by Sierra Oncology and GSK at the time the analysis was conducted; holds stock with Gilead Sciences, GSK. Hiroshi Kosugi has no COIs to declare.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

