NKG2D-bispecific enhances NK and CD8+ T cell antitumor immunity
- PMID: 39112670
- PMCID: PMC11306676
- DOI: 10.1007/s00262-024-03795-2
NKG2D-bispecific enhances NK and CD8+ T cell antitumor immunity
Abstract
Background: Cancer immunotherapy approaches that elicit immune cell responses, including T and NK cells, have revolutionized the field of oncology. However, immunosuppressive mechanisms restrain immune cell activation within solid tumors so additional strategies to augment activity are required.
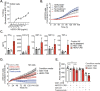
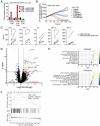
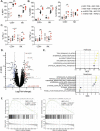
Methods: We identified the co-stimulatory receptor NKG2D as a target based on its expression on a large proportion of CD8+ tumor infiltrating lymphocytes (TILs) from breast cancer patient samples. Human and murine surrogate NKG2D co-stimulatory receptor-bispecifics (CRB) that bind NKG2D on NK and CD8+ T cells as well as HER2 on breast cancer cells (HER2-CRB) were developed as a proof of concept for targeting this signaling axis in vitro and in vivo.
Results: HER2-CRB enhanced NK cell activation and cytokine production when co-cultured with HER2 expressing breast cancer cell lines. HER2-CRB when combined with a T cell-dependent-bispecific (TDB) antibody that synthetically activates T cells by crosslinking CD3 to HER2 (HER2-TDB), enhanced T cell cytotoxicity, cytokine production and in vivo antitumor activity. A mouse surrogate HER2-CRB (mHER2-CRB) improved in vivo efficacy of HER2-TDB and augmented NK as well as T cell activation, cytokine production and effector CD8+ T cell differentiation.
Conclusion: We demonstrate that targeting NKG2D with bispecific antibodies (BsAbs) is an effective approach to augment NK and CD8+ T cell antitumor immune responses. Given the large number of ongoing clinical trials leveraging NK and T cells for cancer immunotherapy, NKG2D-bispecifics have broad combinatorial potential.
Keywords: Bispecifics; CD8+ T cells; NK cells; NKG2D; Synthetic immunity.
© 2024. The Author(s).
Conflict of interest statement
Authors employed by Genentech declare that they have ownership interests in and/or patents with Genentech.
Figures



References
-
- Scheuer W, Friess T, Burtscher H, Bossenmaier B, Endl J, Hasmann M (2009) Strongly enhanced antitumor activity of trastuzumab and pertuzumab combination treatment on HER2-positive human xenograft tumor models. Cancer Res 69:9330–9336. 10.1158/0008-5472.CAN-08-4597 10.1158/0008-5472.CAN-08-4597 - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

