Data Collection and Management of mHealth, Wearables, and Internet of Things in Digital Behavioral Health Interventions With the Awesome Data Acquisition Method (ADAM): Development of a Novel Informatics Architecture
- PMID: 39113371
- PMCID: PMC11322796
- DOI: 10.2196/50043
Data Collection and Management of mHealth, Wearables, and Internet of Things in Digital Behavioral Health Interventions With the Awesome Data Acquisition Method (ADAM): Development of a Novel Informatics Architecture
Abstract
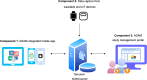
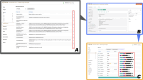
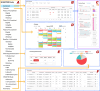
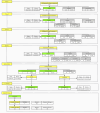
The integration of health and activity data from various wearable devices into research studies presents technical and operational challenges. The Awesome Data Acquisition Method (ADAM) is a versatile, web-based system that was designed for integrating data from various sources and managing a large-scale multiphase research study. As a data collecting system, ADAM allows real-time data collection from wearable devices through the device's application programmable interface and the mobile app's adaptive real-time questionnaires. As a clinical trial management system, ADAM integrates clinical trial management processes and efficiently supports recruitment, screening, randomization, data tracking, data reporting, and data analysis during the entire research study process. We used a behavioral weight-loss intervention study (SMARTER trial) as a test case to evaluate the ADAM system. SMARTER was a randomized controlled trial that screened 1741 participants and enrolled 502 adults. As a result, the ADAM system was efficiently and successfully deployed to organize and manage the SMARTER trial. Moreover, with its versatile integration capability, the ADAM system made the necessary switch to fully remote assessments and tracking that are performed seamlessly and promptly when the COVID-19 pandemic ceased in-person contact. The remote-native features afforded by the ADAM system minimized the effects of the COVID-19 lockdown on the SMARTER trial. The success of SMARTER proved the comprehensiveness and efficiency of the ADAM system. Moreover, ADAM was designed to be generalizable and scalable to fit other studies with minimal editing, redevelopment, and customization. The ADAM system can benefit various behavioral interventions and different populations.
Keywords: Fitbit; Internet of Things; IoT; IoT integration; Nokia; behavioral; clinical trial management; data analysis; data collection; device; integrated system; mHealth; mHealth Fitbit; management; mobile health; remote assessment; research study management; study tracking; tracking; wearable; wearable devices.
© I Wayan Pulantara, Yuhan Wang, Lora E Burke, Susan M Sereika, Zhadyra Bizhanova, Jacob K Kariuki, Jessica Cheng, Britney Beatrice, India Loar, Maribel Cedillo, Molly B Conroy, Bambang Parmanto. Originally published in JMIR mHealth and uHealth (https://mhealth.jmir.org).
Conflict of interest statement
Figures


References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

