Thermo-responsive aqueous two-phase system for two-level compartmentalization
- PMID: 39117632
- PMCID: PMC11310206
- DOI: 10.1038/s41467-024-51043-z
Thermo-responsive aqueous two-phase system for two-level compartmentalization
Abstract
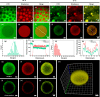
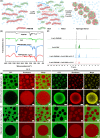
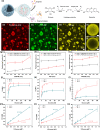
Hierarchical compartmentalization responding to changes in intracellular and extracellular environments is ubiquitous in living eukaryotic cells but remains a formidable task in synthetic systems. Here we report a two-level compartmentalization approach based on a thermo-responsive aqueous two-phase system (TR-ATPS) comprising poly(N-isopropylacrylamide) (PNIPAM) and dextran (DEX). Liquid membraneless compartments enriched in PNIPAM are phase-separated from the continuous DEX solution via liquid-liquid phase separation at 25 °C and shrink dramatically with small second-level compartments generated at the interface, resembling the structure of colloidosome, by increasing the temperature to 35 °C. The TR-ATPS can store biomolecules, program the spatial distribution of enzymes, and accelerate the overall biochemical reaction efficiency by nearly 7-fold. The TR-ATPS inspires on-demand, stimulus-triggered spatiotemporal enrichment of biomolecules via two-level compartmentalization, creating opportunities in synthetic biology and biochemical engineering.
© 2024. The Author(s).
Conflict of interest statement
H.C.S. is a scientific advisor of EN Technology Limited, MicroDiagnostics Limited and Upgrade Biopolymers Limited in which he owns some equity, and also a managing director of the research center, namely Advanced Biomedical Instrumentation Center Limited. The works in the paper are however not directly related to the works of these entities. The remaining authors declare no competing interests.
Figures



References
MeSH terms
Substances
Grants and funding
- No. N_HKU718/19/National Natural Science Foundation of China (National Science Foundation of China)
- 52003287/National Natural Science Foundation of China (National Science Foundation of China)
- 22302128/National Natural Science Foundation of China (National Science Foundation of China)
- Nos. 17317322, 17306221, 17306820, C7165-20GF/Research Grants Council, University Grants Committee (RGC, UGC)
LinkOut - more resources
Full Text Sources

