Extended shortwave infrared absorbing antiaromatic fluorenium-indolizine chromophores
- PMID: 39118622
- PMCID: PMC11304523
- DOI: 10.1039/d4sc00733f
Extended shortwave infrared absorbing antiaromatic fluorenium-indolizine chromophores
Abstract
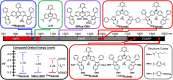
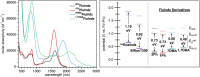
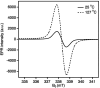
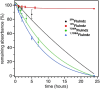
Shortwave infrared (SWIR, 1000-1700 nm) and extended SWIR (ESWIR, 1700-2700 nm) absorbing materials are valuable for applications including fluorescence based biological imaging, photodetectors, and light emitting diodes. Currently, ESWIR absorbing materials are largely dominated by inorganic semiconductors which are often costly both in raw materials and manufacturing processes used to produce them. The development of ESWIR absorbing organic molecules is thus of interest due to the tunability, solution processability, and low cost of organic materials compared to their inorganic counterparts. Herein, through the combination of heterocyclic indolizine donors and an antiaromatic fluorene core, a series of organic chromophores with absorption maxima ranging from 1470-2088 nm (0.84-0.59 eV) and absorption onsets ranging from 1693-2596 nm (0.73-0.48 eV) are designed and synthesized. The photophysical and electrochemical properties of these chromophores, referred to as FluIndz herein, are described via absorption spectroscopy in 17 solvents, cyclic voltammetry, solution photostability, and transient absorption spectroscopy. Molecular orbital energies, predicted electronic transitions, and antiaromaticity are compared to higher energy absorbing chromophores using density functional theory. The presence of thermally accessible diradical states is demonstrated using density functional theory and EPR spectroscopy, while XRD crystallography confirms structural connectivity and existence as a single molecule. Overall, the FluIndz chromophore scaffold exhibits a rational means to access organic chromophores with extremely narrow optical gaps.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures




References
-
- Li H. Wang X. Li X. Zeng S. Chen G. Chem. Mater. 2020;32:3365–3375. doi: 10.1021/acs.chemmater.9b04784. - DOI
-
- Barton J. B., Demro J. C., Amber R., Gasparian G. and Lange M., Defense Public Release: Technical Report, ADA399438, 1998
-
- Wen H. Bellotti E. J. Appl. Phys. 2016;119:205702. doi: 10.1063/1.4951708. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

