Chronic stress deficits in reward behaviour co-occur with low nucleus accumbens dopamine activity during reward anticipation specifically
- PMID: 39123076
- PMCID: PMC11316117
- DOI: 10.1038/s42003-024-06658-9
Chronic stress deficits in reward behaviour co-occur with low nucleus accumbens dopamine activity during reward anticipation specifically
Abstract
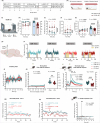
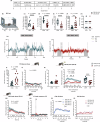
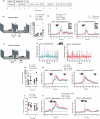
Whilst reward pathologies are major and common in stress-related neuropsychiatric disorders, their neurobiology and treatment are poorly understood. Imaging studies in human reward pathology indicate attenuated BOLD activity in nucleus accumbens (NAc) coincident with reward anticipation but not reinforcement; potentially, this is dopamine (DA) related. In mice, chronic social stress (CSS) leads to reduced reward learning and motivation. Here, DA-sensor fibre photometry is used to investigate whether these behavioural deficits co-occur with altered NAc DA activity during reward anticipation and/or reinforcement. In CSS mice relative to controls: (1) Reduced discriminative learning of the sequence, tone-on + appetitive behaviour = tone-on + sucrose reinforcement, co-occurs with attenuated NAc DA activity throughout tone-on and sucrose reinforcement. (2) Reduced motivation during the sequence, operant behaviour = tone-on + sucrose delivery + sucrose reinforcement, co-occurs with attenuated NAc DA activity at tone-on and typical activity at sucrose reinforcement. (3) Reduced motivation during the sequence, operant behaviour = appetitive behaviour + sociosexual reinforcement, co-occurs with typical NAc DA activity at female reinforcement. Therefore, in CSS mice, low NAc DA activity co-occurs with low reward anticipation and could account for deficits in learning and motivation, with important implications for understanding human reward pathology.
© 2024. The Author(s).
Conflict of interest statement
G.A-L. and B.H. are employees of Boehringer Ingelheim Pharma GmbH & Co KG. C.R.P. has received funding from Boehringer Ingelheim Pharma GmbH & Co KG. All other authors report no biomedical financial interests or potential competing interests.
Figures

Similar articles
-
Orphan receptor-GPR52 inverse agonist efficacy in ameliorating chronic stress-related deficits in reward motivation and phasic accumbal dopamine activity in mice.Transl Psychiatry. 2024 Sep 7;14(1):363. doi: 10.1038/s41398-024-03081-w. Transl Psychiatry. 2024. PMID: 39242529 Free PMC article.
-
Differential Dopamine Release Dynamics in the Nucleus Accumbens Core and Shell Reveal Complementary Signals for Error Prediction and Incentive Motivation.J Neurosci. 2015 Aug 19;35(33):11572-82. doi: 10.1523/JNEUROSCI.2344-15.2015. J Neurosci. 2015. PMID: 26290234 Free PMC article.
-
Conditioned appetitive stimulus increases extracellular dopamine in the nucleus accumbens of the rat.Eur J Neurosci. 2002 Nov;16(10):1987-93. doi: 10.1046/j.1460-9568.2002.02249.x. Eur J Neurosci. 2002. PMID: 12453062
-
Motivational views of reinforcement: implications for understanding the behavioral functions of nucleus accumbens dopamine.Behav Brain Res. 2002 Dec 2;137(1-2):3-25. doi: 10.1016/s0166-4328(02)00282-6. Behav Brain Res. 2002. PMID: 12445713 Review.
-
Effort-related functions of nucleus accumbens dopamine and associated forebrain circuits.Psychopharmacology (Berl). 2007 Apr;191(3):461-82. doi: 10.1007/s00213-006-0668-9. Epub 2007 Jan 16. Psychopharmacology (Berl). 2007. PMID: 17225164 Review.
Cited by
-
Depression in adolescence and young adulthood: the difficulty to integrate motivational/emotional systems.Front Psychol. 2025 Jan 6;15:1391664. doi: 10.3389/fpsyg.2024.1391664. eCollection 2024. Front Psychol. 2025. PMID: 39834756 Free PMC article. Review.
-
The Effects of a Single Vagus Nerve's Neurodynamics on Heart Rate Variability in Chronic Stress: A Randomized Controlled Trial.Sensors (Basel). 2024 Oct 26;24(21):6874. doi: 10.3390/s24216874. Sensors (Basel). 2024. PMID: 39517768 Free PMC article. Clinical Trial.
-
Orphan receptor-GPR52 inverse agonist efficacy in ameliorating chronic stress-related deficits in reward motivation and phasic accumbal dopamine activity in mice.Transl Psychiatry. 2024 Sep 7;14(1):363. doi: 10.1038/s41398-024-03081-w. Transl Psychiatry. 2024. PMID: 39242529 Free PMC article.
-
Disrupted Neuronal Dynamics of Reward Encoding in the Medial Prefrontal Cortex and the Ventral Tegmental Area after Episodic Social Stress.eNeuro. 2025 Jul 25;12(7):ENEURO.0229-25.2025. doi: 10.1523/ENEURO.0229-25.2025. Print 2025 Jul. eNeuro. 2025. PMID: 40664513 Free PMC article.
-
Ethanol-Induced Depression: Exploring the Underlying Molecular Mechanisms.Cell Mol Neurobiol. 2025 May 22;45(1):49. doi: 10.1007/s10571-025-01569-7. Cell Mol Neurobiol. 2025. PMID: 40405002 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

