Recent Advances in the Research on Luotonins A, B, and E
- PMID: 39124927
- PMCID: PMC11314610
- DOI: 10.3390/molecules29153522
Recent Advances in the Research on Luotonins A, B, and E
Abstract
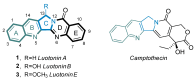
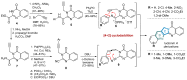
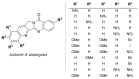
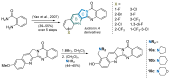
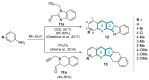
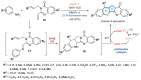
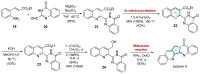
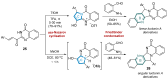
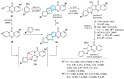
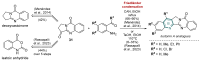
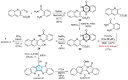
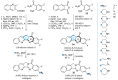
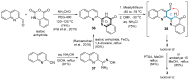
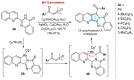
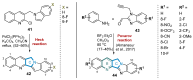
This digest review summarises the most recent progress in the study on luotonins A, B and E. The literature covered in this overview spans from January 2012 to April 2024 and presents synthetic methodologies for the assembly of the quinolinopyrrolo-quinazoline scaffold, the structural motifs present in luotonins A, B, and E, and the evaluation of the biological activities of their derivatives and structural analogues.
Keywords: alkaloids; biological activity; cytotoxicity; luotonins A, B, E; quinolinopyrroloquinazoline.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

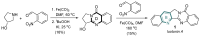
Similar articles
-
Recent advances in the studies on luotonins.Molecules. 2011 Jun 14;16(6):4861-83. doi: 10.3390/molecules16064861. Molecules. 2011. PMID: 21677601 Free PMC article. Review.
-
Novel quinazoline-quinoline alkaloids with cytotoxic and DNA topoisomerase II inhibitory activities.Bioorg Med Chem Lett. 2004 Mar 8;14(5):1193-6. doi: 10.1016/j.bmcl.2003.12.048. Bioorg Med Chem Lett. 2004. PMID: 14980663
-
Recent research progress and outlook in agricultural chemical discovery based on quinazoline scaffold.Pestic Biochem Physiol. 2022 Jun;184:105122. doi: 10.1016/j.pestbp.2022.105122. Epub 2022 May 15. Pestic Biochem Physiol. 2022. PMID: 35715060 Review.
-
Bestowal of Quinazoline Scaffold in Anticancer Drug Discovery.Anticancer Agents Med Chem. 2021;21(11):1350-1368. doi: 10.2174/1871520620666200627205321. Anticancer Agents Med Chem. 2021. PMID: 32593282 Review.
-
Alkaloids and phenylpropanoids from Peganum nigellastrum.Phytochemistry. 2000 Apr;53(8):1075-8. doi: 10.1016/s0031-9422(99)00440-9. Phytochemistry. 2000. PMID: 10820833
Cited by
-
4(3H)-Quinazolinone: A Natural Scaffold for Drug and Agrochemical Discovery.Int J Mol Sci. 2025 Mar 10;26(6):2473. doi: 10.3390/ijms26062473. Int J Mol Sci. 2025. PMID: 40141117 Free PMC article. Review.
References
-
- Nomura T., Ma Z.-Z., Hano Y., Chen Y.-J. Two New Pyrroloquinazolinoquinoline Alkaloids from Peganum nigellastrum. Heterocycles. 1997;46:541–546. doi: 10.3987/COM-97-S65. - DOI
-
- Nomura T., Ma Z.-Z., Hano Y., Chen Y.-J. Two New Quinazoline-Quinoline Alkaloids from Peganum nigellastrum. Heterocycles. 1999;51:1883–1889. doi: 10.3987/COM-99-8595. - DOI
-
- Ma Z., Hano Y., Qiu F., Shao G., Chen Y., Nomura T. Triterpenoids and Alkaloids from the Roots of Peganum Nigellastrum. Nat. Prod. Commun. 2008;3:149–154. doi: 10.1177/1934578X0800300208. - DOI
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

