Functionalizing Thiosemicarbazones for Covalent Conjugation
- PMID: 39125087
- PMCID: PMC11314635
- DOI: 10.3390/molecules29153680
Functionalizing Thiosemicarbazones for Covalent Conjugation
Abstract
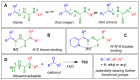
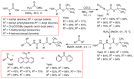
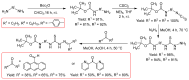
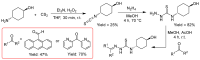
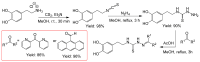
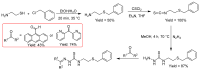
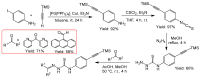
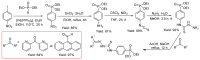
Thiosemicarbazones (TSCs) with their modular character (thiosemicarbazides + carbonyl compound) allow broad variation of up to four substituents on the main R1R2C=N(1)-NH-C(S)-N(4)R3R4 core and are thus interesting tools for the formation of conjugates or the functionalization of nanoparticles (NPs). In this work, di-2-pyridyl ketone was introduced for the coordination of metals and 9-anthraldehyde for luminescence as R1 and R2 to TSCs. R3 and R4 substituents were varied for the formation of conjugates. Amino acids were introduced at the N4 position to produce [R1R2TSC-spacer-amino acid] conjugates. Further, functions such as phosphonic acid (R-P(O)(OH)2), D-glucose, o-hydroquinone, OH, and thiol (SH) were introduced at the N4 position producing [R1R2TSC-spacer-anchor group] conjugates for direct NP anchoring. Phenyl, cyclohexyl, benzyl, ethyl and methyl were used as spacer units. Both phenyl phosphonic acid TSC derivatives were bound on TiO2 NPs as a first example of direct NP anchoring. [R1R2TSC-spacer-end group] conjugates including OH, S-Bn (Bn = benzyl), NH-Boc (Boc = tert-butyloxycarbonyl), COOtBu, C≡CH, or N3 end groups were synthesized for potential covalent binding to functional molecules or functionalized NPs through amide, ester, or triazole functions. The synthesis of the thiosemicarbazides H2NNH-C(S)-NR3R4 starting from amines, including amino acids, SCCl2 or CS2, and hydrazine and their condensation with dipyridyl ketone and anthraldehyde led to 34 new TSC derivatives. They were synthesized in up to six steps with overall yields ranging from 10 to 85% and were characterized by a combination of nuclear magnetic resonance spectroscopy and mass spectrometry. UV-vis absorption and photoluminescence spectroscopy allowed us to easily trace the dipyridyl imine and anthracene chromophores.
Keywords: amino acids; functionalization; glucose; o-hydroquinone; phosphonate; thiosemicarbazones.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures










Similar articles
-
Highly selective addition of chiral, sulfonimidoyl substituted bis(allyl)titanium complexes to N-sulfonyl alpha-imino esters: asymmetric synthesis of gamma,delta-unsaturated alpha-amino acids bearing a chiral, electron-withdrawing nucleofuge at the delta-position.J Am Chem Soc. 2002 Jul 3;124(26):7789-800. doi: 10.1021/ja0201799. J Am Chem Soc. 2002. PMID: 12083933
-
Structural and Metal Ion Effects on Human Topoisomerase IIα Inhibition by α-(N)-Heterocyclic Thiosemicarbazones.Chem Res Toxicol. 2019 Jan 22;32(1):90-99. doi: 10.1021/acs.chemrestox.8b00204. Epub 2018 Dec 11. Chem Res Toxicol. 2019. PMID: 30484632
-
Design, synthesis, and metal binding of novel Pseudo- oligopeptides containing two phosphinic acid groups.Biopolymers. 2008 Jan;89(1):72-85. doi: 10.1002/bip.20855. Biopolymers. 2008. PMID: 17910046
-
Microbial/enzymatic synthesis of chiral drug intermediates.Adv Appl Microbiol. 2000;47:33-78. doi: 10.1016/s0065-2164(00)47001-2. Adv Appl Microbiol. 2000. PMID: 12876794 Review.
-
The role of the adenosinergic system in lung fibrosis.Pharmacol Res. 2013 Oct;76:182-9. doi: 10.1016/j.phrs.2013.08.004. Epub 2013 Aug 28. Pharmacol Res. 2013. PMID: 23994158 Review.
Cited by
-
Thiosemicarbazone-Based Compounds: Cancer Cell Inhibitors with Antioxidant Properties.Molecules. 2025 May 7;30(9):2077. doi: 10.3390/molecules30092077. Molecules. 2025. PMID: 40363882 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Miscellaneous

