Outcomes of the Novel Supreme Drug-Eluting Stent in Complex Coronary Lesions: A PIONEER III Substudy
- PMID: 39130138
- PMCID: PMC11308031
- DOI: 10.1016/j.jscai.2021.100004
Outcomes of the Novel Supreme Drug-Eluting Stent in Complex Coronary Lesions: A PIONEER III Substudy
Abstract
Background: The Supreme healing-targeted drug-eluting stent (DES) is designed to promote endothelial healing to reduce stent-related adverse events. This may be particularly relevant among complex lesions that have a higher rate of adverse events. We sought to compare 1-year outcomes of percutaneous coronary intervention in complex lesions between the Supreme DES and contemporary durable-polymer, everolimus-eluting stents (DP-EES).
Methods: PIONEER III was a multicenter, prospective, single-blind clinical trial, randomizing 1629 patients with either an acute or chronic coronary syndrome in a 2:1 ratio to the Supreme DES or DP-EES. Complex lesions (American College of Cardiology/American Heart Association type B2/C) were found in 1137 patients. Outcomes were also compared for specific parameters of lesion complexity: severe calcification, long length (>20 mm), and severe tortuosity. The primary end point was target lesion failure at 1 year.
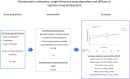
Results: At 1 year, there was no difference in target lesion failure between the Supreme DES and DP-EES: (5.7% vs 5.6%; hazard ratio 1.00, 95% confidence interval 0.59-1.68, P = .99). Similarly, there were no differences in the secondary end points of lesion success (99.7% vs 99.4%, P = .41), device success (97.0% vs 98.5%, P = .14), target vessel failure (6.5% vs 7.4%, P = .50), major adverse cardiac events (7.8% vs 8.5%, P = .64), or stent thrombosis (0.7% vs 1.1%, P = .48). A trend was observed toward a higher rate of target lesion revascularization with the Supreme DES (2.5% vs 0.9%, P = .06).
Conclusions: This study suggests that the Supreme DES is as effective and safe at 1 year compared with the standard DP-EES across a broad spectrum of lesion complexity.
Keywords: Acute coronary syndrome; Coronary artery disease; Coronary revascularization; Drug-eluting stent; Percutaneous coronary intervention.
Crown Copyright © 2021 Published by Elsevier Inc. on behalf of the Society for Cardiovascular Angiography and Interventions Foundation.
Figures


References
-
- Sarno G., Lagerqvist B., Fröbert O., et al. Lower risk of stent thrombosis and restenosis with unrestricted use of ‘new-generation’ drug-eluting stents: a report from the nationwide Swedish Coronary Angiography and Angioplasty Registry (SCAAR) Eur Heart J. 2012;33:606–613. - PubMed
-
- Bangalore S., Kumar S., Fusaro M., et al. Short-and long-term outcomes with drug-eluting and bare-metal coronary stents: a mixed-treatment comparison analysis of 117 762 patient-years of follow-up from randomized trials. Circulation. 2012;125:2873–2891. - PubMed
-
- Lansky A., Kereiakes D., Baumbach A., et al. Novel Supreme drug-eluting stents with early synchronized antiproliferative drug delivery to Inhibit smooth muscle cell proliferation after drug-eluting stents implantation in coronary artery disease: results of the PIONEER III randomized clinical trial. Circulation. 2021;143:2143–2154. - PubMed
-
- Beale C.E., Abbott J.D. Drug eluting stents for very long lesions: go long, but know the risks. Catheter Cardiovasc Interv. 2017:992–993. - PubMed
-
- Geraci S., Kawamoto H., Caramanno G., et al. Bioresorbable everolimus-eluting vascular scaffold for long coronary lesions: a subanalysis of the international, multicenter GHOST-EU registry. JACC Cardiovasc Interv. 2017;10:560–568. - PubMed
LinkOut - more resources
Full Text Sources

