Risk of Bleeding Among Cangrelor-Treated Patients Administered Upstream P2Y12 Inhibitor Therapy: The CAMEO Registry
- PMID: 39132213
- PMCID: PMC11308101
- DOI: 10.1016/j.jscai.2023.101202
Risk of Bleeding Among Cangrelor-Treated Patients Administered Upstream P2Y12 Inhibitor Therapy: The CAMEO Registry
Erratum in
-
Correction.J Soc Cardiovasc Angiogr Interv. 2025 Aug 9;4(10Part A):103930. doi: 10.1016/j.jscai.2025.103930. eCollection 2025 Oct. J Soc Cardiovasc Angiogr Interv. 2025. PMID: 41268087 Free PMC article.
Abstract
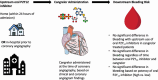
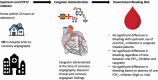
Background: Little is known about the bleeding risk associated with cangrelor use in patients with myocardial infarction (MI) who are exposed to an oral P2Y12 inhibitor before coronary angiography.
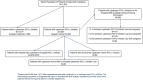
Methods: Cangrelor in Acute MI: Effectiveness and Outcomes (CAMEO) is an observational registry studying platelet inhibition for patients with MI. Upstream oral P2Y12 inhibition was defined as receipt of an oral P2Y12 inhibitor within 24 hours before hospitalization or in-hospital before angiography. Among cangrelor-treated patients, we compared bleeding after cangrelor use through 7 days postdischarge between patients with and without upstream oral P2Y12 inhibitor exposure.
Results: Among 1802 cangrelor-treated patients with MI, 385 (21.4%) received upstream oral P2Y12 inhibitor treatment. Of these, 101 patients (33.8%) started cangrelor within 1 hour, 103 (34.4%) between 1 and 3 hours, and 95 (31.8%), >3 hours after in-hospital oral P2Y12 inhibitor administration; the remaining received an oral P2Y12 inhibitor before hospitalization. There was no statistically significant difference in rates of bleeding among cangrelor-treated patients with and without upstream oral P2Y12 inhibitor exposure (6.5% vs 8.8%; adjusted odds ratio [OR], 0.62; 95% CI, 0.38-1.01). Bleeding was observed in 5.0%, 10.7%, and 3.2% of patients treated with cangrelor <1, 1 to 3, and >3 hours after the last oral PY12 inhibitor dose, respectively; bleeding rates were not statistically different between groups (1-3 hours vs <1 hour: adjusted OR, 2.70; 95% CI, 0.87-8.32; >3 hours vs <1 hour: adjusted OR, 0.65; 95% CI, 0.15-2.85).
Conclusions: Bleeding risk was not observed to be significantly higher after cangrelor treatment in patients with and without upstream oral P2Y12 inhibitor exposure.
Keywords: P2Y12 inhibitor; bleeding; cangrelor; myocardial infarction.
© 2023 The Author(s).
Figures

References
-
- Bhatt D.L., Stone G.W., Mahaffey K.W., et al. Effect of platelet inhibition with cangrelor during PCI on ischemic events. N Engl J Med. 2013;368(14):1303–1313. - PubMed
-
- Lawton J.S., Tamis-Holland J.E., Bangalore S., et al. 2021 ACC/AHA/SCAI guideline for coronary artery revascularization: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145(3):e18–e114. - PubMed
-
- Franchi F., Rollini F., Angiolillo D.J. Antithrombotic therapy for patients with STEMI undergoing primary PCI. Nat Rev Cardiol. 2017;14(6):361–379. - PubMed
-
- Capodanno D., Angiolillo D.J. Timing, selection, modulation, and duration of P2Y12 inhibitors for patients with acute coronary syndromes undergoing PCI. J Am Coll Cardiol Interv. 2023;16(1):1–18. - PubMed
LinkOut - more resources
Full Text Sources
Medical

