Bioengineering-tissue strategies to model mammalian implantation in vitro
- PMID: 39132254
- PMCID: PMC11310004
- DOI: 10.3389/fbioe.2024.1430235
Bioengineering-tissue strategies to model mammalian implantation in vitro
Abstract
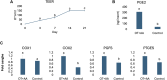
During mammalian implantation, complex and well-orchestrated interactions between the trophectoderm of implanting blastocysts and the maternal endometrium lead to a successful pregnancy. On the other hand, alteration in endometrium-blastocyst crosstalk often causes implantation failure, pregnancy loss, and complications that result in overall infertility. In domestic animals, this represents one of the major causes of economic losses and the understanding of the processes taking place during the early phases of implantation, in both healthy and pathological conditions, is of great importance, to enhance livestock system efficiency. Here we develop highly predictive and reproducible functional tridimensional (3D) in vitro models able to mimic the two main actors that play a key role at this developmental stage: the blastocyst and the endometrium. In particular, we generate a 3D endometrial model by co-culturing primary epithelial and stromal cells, isolated from sow uteri, onto highly porous polystyrene scaffolds. In parallel, we chemically reprogram porcine adult dermal fibroblasts and encapsulate them into micro-bioreactors to create trophoblast (TR) spheroids. Finally, we combine the generated artificial endometrium with the TR spheroids to model mammalian implantation in vitro and mimic the embryo-maternal interactions. The protocols here described allow the generation of reproducible and functional 3D models of both the maternal compartment as well as the implanting embryo, able to recreate in vitro the architecture and physiology of the two tissues in vivo. We suggest that these models can find useful applications to further elucidate early implantation mechanisms and to study the complex interactions between the maternal tissue and the developing embryos.
Keywords: 3D endometrial model; chemical reprogramming; microbioreactors; scaffolds; trophoblast spheroid.
Copyright © 2024 Pennarossa, Arcuri, Zmijewska, Orini, Gandolfi and Brevini.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



References
-
- Assheton R. (1906). “The morphology of the ungulate placenta, particularly the development of that organ in the sheep, and notes upon the placenta of the elephant and hyrax on JSTOR,” in Philosophical transactions of the royal society of London. series B, containing papers of a biological character (London: JSTOR; ), 143–220. Available at: https://www.jstor.org/stable/91945?seq=1 (Accessed May 4, 2021).

