Enhancing radiotherapy response via intratumoral injection of a TLR9 agonist in autochthonous murine sarcomas
- PMID: 39133651
- PMCID: PMC11383182
- DOI: 10.1172/jci.insight.178767
Enhancing radiotherapy response via intratumoral injection of a TLR9 agonist in autochthonous murine sarcomas
Abstract
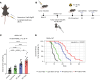
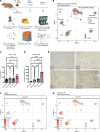
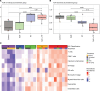
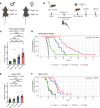
Radiation therapy (RT) is frequently used to treat cancers, including soft-tissue sarcomas. Prior studies established that the toll-like receptor 9 (TLR9) agonist cytosine-phosphate-guanine oligodeoxynucleotide (CpG) enhances the response to RT in transplanted tumors, but the mechanisms of this enhancement remain unclear. Here, we used CRISPR/Cas9 and the chemical carcinogen 3-methylcholanthrene (MCA) to generate autochthonous soft-tissue sarcomas with high tumor mutation burden. Treatment with a single fraction of 20 Gy RT and 2 doses of CpG significantly enhanced tumor response, which was abrogated by genetic or immunodepletion of CD8+ T cells. To characterize the immune response to CpG+RT, we performed bulk RNA-Seq, single-cell RNA-Seq, and mass cytometry. Sarcomas treated with 20 Gy and CpG demonstrated increased CD8 T cells expressing markers associated with activation and proliferation, such as Granzyme B, Ki-67, and IFN-γ. CpG+RT also upregulated antigen presentation pathways on myeloid cells. Furthermore, in sarcomas treated with CpG+RT, TCR clonality analysis suggests an increase in clonal T cell dominance. Collectively, these findings demonstrate that CpG+RT significantly delays tumor growth in a CD8 T cell-dependent manner. These results provide a strong rationale for clinical trials evaluating CpG or other TLR9 agonists with RT in patients with soft-tissue sarcoma.
Keywords: Cancer immunotherapy; Immunology; Oncology; Radiation therapy; T cells.
Figures


Update of
-
Enhancing radiotherapy response via intratumoral injection of the TLR9 agonist CpG to stimulate CD8 T cells in an autochthonous mouse model of sarcoma.bioRxiv [Preprint]. 2024 Jan 4:2024.01.03.573968. doi: 10.1101/2024.01.03.573968. bioRxiv. 2024. Update in: JCI Insight. 2024 Jun 13;9(14):e178767. doi: 10.1172/jci.insight.178767. PMID: 38260522 Free PMC article. Updated. Preprint.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

