Convergent Comodulation Reduces Interindividual Variability of Circuit Output
- PMID: 39134416
- PMCID: PMC11403100
- DOI: 10.1523/ENEURO.0167-24.2024
Convergent Comodulation Reduces Interindividual Variability of Circuit Output
Abstract
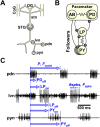
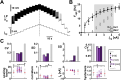
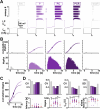
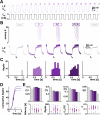
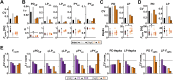
Ionic current levels of identified neurons vary substantially across individual animals. Yet, under similar conditions, neural circuit output can be remarkably similar, as evidenced in many motor systems. All neural circuits are influenced by multiple neuromodulators, which provide flexibility to their output. These neuromodulators often overlap in their actions by modulating the same channel type or synapse, yet have neuron-specific actions resulting from distinct receptor expression. Because of this different receptor expression pattern, in the presence of multiple convergent neuromodulators, a common downstream target would be activated more uniformly in circuit neurons across individuals. We therefore propose that a baseline tonic (non-saturating) level of comodulation by convergent neuromodulators can reduce interindividual variability of circuit output. We tested this hypothesis in the pyloric circuit of the crab, Cancer borealis Multiple excitatory neuropeptides converge to activate the same voltage-gated current in this circuit, but different subsets of pyloric neurons have receptors for each peptide. We quantified the interindividual variability of the unmodulated pyloric circuit output by measuring the activity phases, cycle frequency, and intraburst spike number and frequency. We then examined the variability in the presence of different combinations and concentrations of three neuropeptides. We found that at mid-level concentration (30 nM) but not at near-threshold (1 nM) or saturating (1 µM) concentrations, comodulation by multiple neuropeptides reduced the circuit output variability. Notably, the interindividual variability of response properties of an isolated neuron was not reduced by comodulation, suggesting that the reduction of output variability may emerge as a network effect.
Keywords: central pattern generator; neuromodulation; stomatogastric; variability.
Copyright © 2024 Schneider et al.
Conflict of interest statement
The authors declare no competing financial interests.
Figures

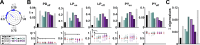
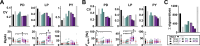
Update of
-
Comodulation reduces interindividual variability of circuit output.bioRxiv [Preprint]. 2023 Aug 13:2023.06.03.543573. doi: 10.1101/2023.06.03.543573. bioRxiv. 2023. Update in: eNeuro. 2024 Sep 10;11(9):ENEURO.0167-24.2024. doi: 10.1523/ENEURO.0167-24.2024. PMID: 37383946 Free PMC article. Updated. Preprint.
References
-
- Atamturktur S, Nadim F (2011) Effects of neuromodulator RPCH on short-term dynamics of a key synapse in an oscillatory network of crab Cancer borealis. J Appl Biol Sci 5:59–72.
-
- Berens P (2009) Circstat: a MATLAB toolbox for circular statistics. J Stat Softw 31:1–21. 10.18637/jss.v031.i10 - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
