Secoisolariciresinol diglucoside attenuates neuroinflammation and cognitive impairment in female Alzheimer's disease mice via modulating gut microbiota metabolism and GPER/CREB/BDNF pathway
- PMID: 39135052
- PMCID: PMC11320852
- DOI: 10.1186/s12974-024-03195-4
Secoisolariciresinol diglucoside attenuates neuroinflammation and cognitive impairment in female Alzheimer's disease mice via modulating gut microbiota metabolism and GPER/CREB/BDNF pathway
Abstract
Background: Gender is a significant risk factor for late-onset Alzheimer's disease (AD), often attributed to the decline of estrogen. The plant estrogen secoisolariciresinol diglucoside (SDG) has demonstrated anti-inflammatory and neuroprotective effects. However, the protective effects and mechanisms of SDG in female AD remain unclear.
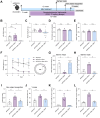
Methods: Ten-month-old female APPswe/PSEN1dE9 (APP/PS1) transgenic mice were treated with SDG to assess its potential ameliorative effects on cognitive impairments in a female AD model through a series of behavioral and biochemical experiments. Serum levels of gut microbial metabolites enterodiol (END) and enterolactone (ENL) were quantified using HPLC-MS. Correlation analysis and broad-spectrum antibiotic cocktail (ABx) treatment were employed to demonstrate the involvement of END and ENL in SDG's cognitive improvement effects in female APP/PS1 mice. Additionally, an acute neuroinflammation model was constructed in three-month-old C57BL/6J mice treated with lipopolysaccharide (LPS) and subjected to i.c.v. injection of G15, an inhibitor of G protein-coupled estrogen receptor (GPER), to investigate the mediating role of the estrogen receptor GPER in the cognitive benefits conferred by SDG.
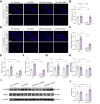
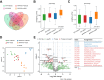
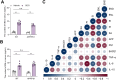
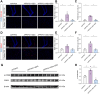
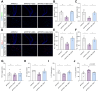
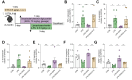
Results: SDG administration resulted in significant improvements in spatial, recognition, and working memory in female APP/PS1 mice. Neuroprotective effects were observed, including enhanced expression of CREB/BDNF and PSD-95, reduced β-amyloid (Aβ) deposition, and decreased levels of TNF-α, IL-6, and IL-10. SDG also altered gut microbiota composition, increasing serum levels of END and ENL. Correlation analysis indicated significant associations between END, ENL, cognitive performance, hippocampal Aβ-related protein mRNA expression, and cortical neuroinflammatory cytokine levels. The removal of gut microbiota inhibited END and ENL production and eliminated the neuroprotective effects of SDG. Furthermore, GPER was found to mediate the inhibitory effects of SDG on neuroinflammatory responses.
Conclusion: These findings suggest that SDG promotes the production of gut microbial metabolites END and ENL, which inhibit cerebral β-amyloid deposition, activate GPER to enhance CREB/BDNF signaling pathways, and suppress neuroinflammatory responses. Consequently, SDG exerts neuroprotective effects and ameliorates cognitive impairments associated with AD in female mice.
Keywords: Alzheimer’s disease; Cognitive impairment; GPER; Gut microbiota; Neuroinflammation; Secoisolariciresinol diglucoside.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



References
-
- Farrer LA, Cupples LA, Haines JL, Hyman B, Kukull WA, Mayeux R, Myers RH, Pericak-Vance MA, Risch N, van Duijn CM. Effects of age, sex, and ethnicity on the association between apolipoprotein E genotype and Alzheimer disease. A meta-analysis. APOE and Alzheimer Disease Meta Analysis Consortium. JAMA. 1997;278:1349–56. 10.1001/jama.1997.03550160069041 - DOI - PubMed
-
- 2016 Alzheimer’s disease facts and figures. Alzheimers Dement 2016, 12:459–509. - PubMed
-
- Uddin MS, Rahman MM, Jakaria M, Rahman MS, Hossain MS, Islam A, Ahmed M, Mathew B, Omar UM, Barreto GE, Ashraf GM. Estrogen Signaling in Alzheimer’s Disease: Molecular insights and therapeutic targets for Alzheimer’s dementia. Mol Neurobiol. 2020;57:2654–70. 10.1007/s12035-020-01911-8 - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

