Paired organoids from primary gastric cancer and lymphatic metastasis are useful for personalized medicine
- PMID: 39135062
- PMCID: PMC11318189
- DOI: 10.1186/s12967-024-05512-0
Paired organoids from primary gastric cancer and lymphatic metastasis are useful for personalized medicine
Abstract
Background: Organoids are approved by the US FDA as an alternative to animal experiments to guide drug development and for sensitivity screening. Stable organoids models of gastric cancer are desirable for personalized medicine and drug screening.
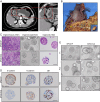
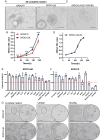
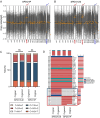
Methods: Tumor tissues from a primary cancer of the stomach and metastatic cancer of the lymph node were collected for 3D culture. By long-term culture for over 50 generations in vitro, we obtained stably growing organoid lines. We analyzed short tandem repeats (STRs) and karyotypes of cancer cells, and tumorigenesis of the organoids in nude mice, as well as multi-omics profiles of the organoids. A CCK8 method was used to determine the drugs sensitivity to fluorouracil (5-Fu), platinum and paclitaxel.
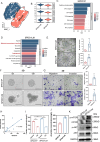
Results: Paired organoid lines from primary cancer (SPDO1P) and metastatic lymph node (SPDO1LM) were established with unique STRs and karyotypes. The organoid lines resulted in tumorigenesis in vivo and had clear genetic profiles. Compared to SPDO1P from primary cancer, upregulated genes of SPDO1LM from the metastatic lymph node were enriched in pathways of epithelial-mesenchymal transition and angiogenesis with stronger abilities of cell migration, invasion, and pro-angiogenesis. Based on drug sensitivity analysis, the SOX regimen (5-Fu plus oxaliplatin) was used for chemotherapy with an optimal clinical outcome.
Conclusions: The organoid lines recapitulate the drug sensitivity of the parental tissues. The paired organoid lines present a step-change toward living biobanks for further translational usage.
Keywords: Drug sensitivity; Gastric cancer; Living biobank; Organoid models; Translational usage.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
PI3 expression predicts recurrence after chemotherapy with DNA-damaging drugs in gastric cancer.J Pathol. 2025 Apr;265(4):472-485. doi: 10.1002/path.6400. Epub 2025 Feb 20. J Pathol. 2025. PMID: 39980125 Free PMC article.
-
Chemotherapy for advanced gastric cancer.Cochrane Database Syst Rev. 2017 Aug 29;8(8):CD004064. doi: 10.1002/14651858.CD004064.pub4. Cochrane Database Syst Rev. 2017. PMID: 28850174 Free PMC article.
-
A rapid and systematic review of the clinical effectiveness and cost-effectiveness of paclitaxel, docetaxel, gemcitabine and vinorelbine in non-small-cell lung cancer.Health Technol Assess. 2001;5(32):1-195. doi: 10.3310/hta5320. Health Technol Assess. 2001. PMID: 12065068
-
Malignant ascites-derived organoid (MADO) cultures for gastric cancer in vitro modelling and drug screening.J Cancer Res Clin Oncol. 2019 Nov;145(11):2637-2647. doi: 10.1007/s00432-019-03004-z. Epub 2019 Oct 9. J Cancer Res Clin Oncol. 2019. PMID: 31598791 Free PMC article.
-
High-Throughput Drug Screening of Clear Cell Ovarian Cancer Organoids Reveals Vulnerability to Proteasome Inhibitors and Dinaciclib and Identifies AGR2 as a Therapeutic Target.Cancer Res Commun. 2025 Jun 1;5(6):1018-1033. doi: 10.1158/2767-9764.CRC-25-0024. Cancer Res Commun. 2025. PMID: 40459063 Free PMC article.
Cited by
-
The application of organoids in investigating immune evasion in the microenvironment of gastric cancer and screening novel drug candidates.Mol Cancer. 2025 Apr 26;24(1):125. doi: 10.1186/s12943-025-02328-4. Mol Cancer. 2025. PMID: 40287758 Free PMC article. Review.
-
Standard: Human gastric cancer organoids.Cell Regen. 2024 Dec 27;13(1):33. doi: 10.1186/s13619-024-00217-7. Cell Regen. 2024. PMID: 39729207 Free PMC article.
-
Living biobank: Standardization of organoid construction and challenges.Chin Med J (Engl). 2024 Dec 20;137(24):3050-3060. doi: 10.1097/CM9.0000000000003414. Epub 2024 Dec 12. Chin Med J (Engl). 2024. PMID: 39663560 Free PMC article. Review.
-
Standard: Human gastric organoids.Cell Regen. 2025 Jan 14;14(1):2. doi: 10.1186/s13619-024-00218-6. Cell Regen. 2025. PMID: 39808364 Free PMC article.
-
Factors associated with actionable gene aberrations in pancreatic cancer based on the C-CAT database.J Gastroenterol. 2025 Aug;60(8):1026-1035. doi: 10.1007/s00535-025-02253-9. Epub 2025 May 2. J Gastroenterol. 2025. PMID: 40314773 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

