Age-dependent changes in phagocytic activity: in vivo response of mouse pulmonary antigen presenting cells to direct lung delivery of charged PEGDA nanoparticles
- PMID: 39135064
- PMCID: PMC11318229
- DOI: 10.1186/s12951-024-02743-7
Age-dependent changes in phagocytic activity: in vivo response of mouse pulmonary antigen presenting cells to direct lung delivery of charged PEGDA nanoparticles
Erratum in
-
Correction: Age-dependent changes in phagocytic activity: in vivo response of mouse pulmonary antigen presenting cells to direct lung delivery of charged PEGDA nanoparticles.J Nanobiotechnology. 2024 Dec 12;22(1):751. doi: 10.1186/s12951-024-03040-z. J Nanobiotechnology. 2024. PMID: 39663532 Free PMC article. No abstract available.
Abstract
Background: Current needle-based vaccination for respiratory viruses is ineffective at producing sufficient, long-lasting local immunity in the elderly. Direct pulmonary delivery to the resident local pulmonary immune cells can create long-term mucosal responses. However, criteria for drug vehicle design rules that can overcome age-specific changes in immune cell functions have yet to be established.
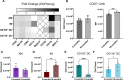
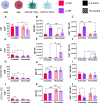
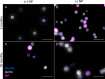
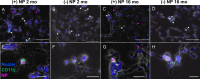
Results: Here, in vivo charge-based nanoparticle (NP) uptake was compared in mice of two age groups (2- and 16-months) within the four notable pulmonary antigen presenting cell (APC) populations: alveolar macrophages (AM), interstitial macrophages (IM), CD103+ dendritic cells (DCs), and CD11b+ DCs. Both macrophage populations exhibited preferential uptake of anionic nanoparticles but showed inverse rates of phagocytosis between the AM and IM populations across age. DC populations demonstrated preferential uptake of cationic nanoparticles, which remarkably did not significantly change in the aged group. Further characterization of cell phenotypes post-NP internalization demonstrated unique surface marker expression and activation levels for each APC population, showcasing heightened DC inflammatory response to NP delivery in the aged group.
Conclusion: The age of mice demonstrated significant preferences in the charge-based NP uptake in APCs that differed greatly between macrophages and DCs. Carefully balance of the targeting and activation of specific types of pulmonary APCs will be critical to produce efficient, age-based vaccines for the growing elderly population.
Keywords: Aging; Antigen presenting cells; Nanoparticles; Phagocytosis; Pulmonary delivery; Pulmonary immunity.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

