Inhibitors identify an auxiliary role for mTOR signalling in necroptosis execution downstream of MLKL activation
- PMID: 39136677
- PMCID: PMC11555701
- DOI: 10.1042/BCJ20240255
Inhibitors identify an auxiliary role for mTOR signalling in necroptosis execution downstream of MLKL activation
Abstract
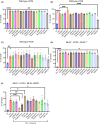
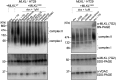
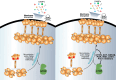
Necroptosis is a lytic and pro-inflammatory form of programmed cell death executed by the terminal effector, the MLKL (mixed lineage kinase domain-like) pseudokinase. Downstream of death and Toll-like receptor stimulation, MLKL is trafficked to the plasma membrane via the Golgi-, actin- and microtubule-machinery, where activated MLKL accumulates until a critical lytic threshold is exceeded and cell death ensues. Mechanistically, MLKL's lytic function relies on disengagement of the N-terminal membrane-permeabilising four-helix bundle domain from the central autoinhibitory brace helix: a process that can be experimentally mimicked by introducing the R30E MLKL mutation to induce stimulus-independent cell death. Here, we screened a library of 429 kinase inhibitors for their capacity to block R30E MLKL-mediated cell death, to identify co-effectors in the terminal steps of necroptotic signalling. We identified 13 compounds - ABT-578, AR-A014418, AZD1480, AZD5363, Idelalisib, Ipatasertib, LJI308, PHA-793887, Rapamycin, Ridaforolimus, SMI-4a, Temsirolimus and Tideglusib - each of which inhibits mammalian target of rapamycin (mTOR) signalling or regulators thereof, and blocked constitutive cell death executed by R30E MLKL. Our study implicates mTOR signalling as an auxiliary factor in promoting the transport of activated MLKL oligomers to the plasma membrane, where they accumulate into hotspots that permeabilise the lipid bilayer to cause cell death.
Keywords: mechanistic target of rapamycin; necroptosis; protein kinase; pseudokinase; signalling.
© 2024 The Author(s).
Conflict of interest statement
All authors contribute, or have contributed, to a project developing necroptosis pathway inhibitors in collaboration with Anaxis Pharma Pty Ltd. J.M.M. has received research funding from Anaxis Pharma Pty Ltd.
Figures



Similar articles
-
The brace helices of MLKL mediate interdomain communication and oligomerisation to regulate cell death by necroptosis.Cell Death Differ. 2018 Sep;25(9):1567-1580. doi: 10.1038/s41418-018-0061-3. Epub 2018 Feb 14. Cell Death Differ. 2018. PMID: 29445128 Free PMC article.
-
Death at a funeral: Activation of the dead enzyme, MLKL, to kill cells by necroptosis.Curr Opin Struct Biol. 2024 Oct;88:102891. doi: 10.1016/j.sbi.2024.102891. Epub 2024 Jul 25. Curr Opin Struct Biol. 2024. PMID: 39059047 Review.
-
The Lck inhibitor, AMG-47a, blocks necroptosis and implicates RIPK1 in signalling downstream of MLKL.Cell Death Dis. 2022 Apr 1;13(4):291. doi: 10.1038/s41419-022-04740-w. Cell Death Dis. 2022. PMID: 35365636 Free PMC article.
-
Activation of the pseudokinase MLKL unleashes the four-helix bundle domain to induce membrane localization and necroptotic cell death.Proc Natl Acad Sci U S A. 2014 Oct 21;111(42):15072-7. doi: 10.1073/pnas.1408987111. Epub 2014 Oct 6. Proc Natl Acad Sci U S A. 2014. PMID: 25288762 Free PMC article.
-
The Role of the Key Effector of Necroptotic Cell Death, MLKL, in Mouse Models of Disease.Biomolecules. 2021 May 28;11(6):803. doi: 10.3390/biom11060803. Biomolecules. 2021. PMID: 34071602 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

