PCSK9 inhibitors in real life-Cardiometabolic risk management in dyslipidemic patients in Vienna
- PMID: 39136768
- PMCID: PMC12081568
- DOI: 10.1007/s00508-024-02402-9
PCSK9 inhibitors in real life-Cardiometabolic risk management in dyslipidemic patients in Vienna
Abstract
Background: Proprotein convertase subtilisin kexin type 9 (PCSK9) inhibitors have emerged as important therapeutic options for patients unable to achieve the low-density lipoprotein cholesterol (LDL‑C) target or to tolerate alternative lipid-lowering agents. The aim of this study was to investigate the efficacy of PCSK9 inhibitor treatment in tertiary routine care, by determining the percentage of patients reaching individual LDL‑C target levels 1 year after treatment initiation.
Patients and methods: Patients routinely started on PCSK9 inhibitors at our lipid clinic between 2017 and 2020 were retrospectively analyzed. Attainment of the LDL‑C target, utilization of follow-ups, cardiovascular events and effects on laboratory parameters were investigated.
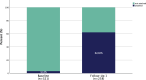
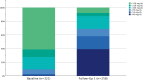
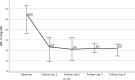
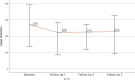
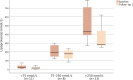
Results: In this study 347 patients were included, with the majority managed in secondary prevention (94.5%). The LDL‑C target was achieved by 44.9% after ca. 14 months, with differences between statin users and non-users (51.0% vs. 22.7%; p < 0.001). The median LDL‑C decreased from 126.00 mg/dL at baseline to 48 mg/dL (-61.6%; -77.00 mg/dL; p < 0.001) after ~2 months and to 60 mg/dL (-52.9%; -59.00 mg/dL; p < 0.001) after ~14 months. Median lipoprotein(a) levels decreased significantly from 184.0 nmol/L to 165.5 nmol/L (-25.9%; -25.5 nmol/L; p = 0.001) after ~2 months, whereas no effects on creatine kinase, amylase and lipase were detectable. Of the patients 15% utilized 4 follow-ups. The PCSK9 inhibitor intolerance occurred in 3.5% of patients.
Conclusion: With the effect of LDL-lowering remaining constant over 14 months, PCSK9 inhibitor treatment showed effective and sustainable LDL‑C lowering in a majority of patients in secondary prevention, bringing them closer to the recommended LDL‑C goal, particularly those under concomitant statin medication. Treatment with PCSK9 inhibitors appears to be well-tolerated, confirming data from clinical trials in real life.
Keywords: Austria; Follow-up; Lipoprotein(a); Real-world data; Target-Attainment.
© 2024. The Author(s).
Conflict of interest statement
Conflict of interest: M. Ferch, C. Sert, P. Fellinger, A. Kautzky-Willer and Y. Winhofer-Stöckl declare that they have no competing interests.
Figures
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

