Safety and feasibility of atrial fibrillation ablation after left atrial appendage closure: A single-center experience of the left atrial appendage closure first strategy
- PMID: 39139871
- PMCID: PMC11317688
- DOI: 10.1002/joa3.13073
Safety and feasibility of atrial fibrillation ablation after left atrial appendage closure: A single-center experience of the left atrial appendage closure first strategy
Abstract
Background: Patients with atrial fibrillation (AF) who are not suitable for long-term anticoagulant therapy undergo percutaneous left atrial appendage closure (LAAC). The safety and feasibility of left atrial catheter ablation (CA) procedures after LAAC remain unclear. This study aimed to clarify the feasibility and safety of CA after LAAC, including in the early phase within 180 days.
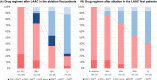
Methods: Characteristics and clinical outcomes of 46 patients with AF who had undergone both CA and LAAC within 2 years (mean age, 72 years; 29 men) were compared between those who had undergone CA-first (31 patients) and LAAC-first (15 patients).
Results: The mean CHA₂DS₂-VASc and HAS-BLED scores were 4.8 and 3.3 points, respectively. The LAAC-first strategy was often used in patients with prior major bleeding and LAA thrombosis or sludge. In the LAAC-first group, the mean duration between both procedures was 212 days, and all LAAC-first patients, including seven patients in the early phase, could undergo CA without LAAC device-related complications; moreover, no cardiovascular adverse events were reported after both procedures (mean periods: 420 days). After CA post-LAAC, no device-related adverse events (device-related thrombosis, new peri-device leak appearance, peri-device leak increase, or device dislodgement) were observed, whereas, after LAAC post-CA, 3 new peri-device leak appearance events and 1 peri-device leak increase event were observed, especially patients who underwent LAAC in the early phase post-CA.
Conclusion: Based on single-center experience, left atrial CA in the presence of an LAAC device implanted including the early phase was safe and feasible.
Keywords: antithrombotic regimen; atrial fibrillation; catheter ablation; left atrial appendage closure; peri‐device leak.
© 2024 The Author(s). Journal of Arrhythmia published by John Wiley & Sons Australia, Ltd on behalf of Japanese Heart Rhythm Society.
Conflict of interest statement
Dr. Kubo is a clinical proctor for Boston Scientific. Dr. Kubo received honorarium from Boston Scientific. All other authors declare no conflict of interest.
Figures


References
-
- Marrouche NF, Brachmann J, Andresen D, Siebels J, Boersma L, Jordaens L, et al. Catheter ablation for atrial fibrillation with heart failure. N Engl J Med. 2018;378:417–427. - PubMed
-
- Tzou WS, Marchlinski FE, Zado ES, Lin D, Dixit S, Callans DJ, et al. Long‐term outcome after successful catheter ablation of atrial fibrillation. Circ Arrhythm Electrophysiol. 2010;3:237–242. - PubMed
-
- Hindricks G, Potpara T, Dagres N, Arbelo E, Bax JJ, Blomström‐Lundqvist C, et al. 2020 ESC guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio‐Thoracic Surgery (EACTS): the task force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) developed with the special contribution of the European heart rhythm association (EHRA) of the ESC. Eur Heart J. 2021;42:373–498. - PubMed
-
- Nogami A, Kurita T, Abe H, Ando K, Ishikawa T, Imai K, et al. JCS/JHRS 2019 guideline on non‐pharmacotherapy of cardiac arrhythmias. Circ J. 2021;85:1104–1244. - PubMed
-
- Holmes DR, Reddy VY, Turi ZG, Doshi SK, Sievert H, Buchbinder M, et al. Percutaneous closure of the left atrial appendage versus warfarin therapy for prevention of stroke in patients with atrial fibrillation: a randomised non‐inferiority trial. Lancet. 2009;374:534–542. - PubMed
LinkOut - more resources
Full Text Sources

