Multimodal neuroimaging to characterize symptom-specific networks in movement disorders
- PMID: 39143114
- PMCID: PMC11324766
- DOI: 10.1038/s41531-024-00774-3
Multimodal neuroimaging to characterize symptom-specific networks in movement disorders
Abstract
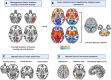
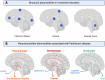
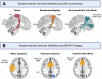
Movement disorders, such as Parkinson's disease, essential tremor, and dystonia, are characterized by their predominant motor symptoms, yet diseases causing abnormal movement also encompass several other symptoms, including non-motor symptoms. Here we review recent advances from studies of brain lesions, neuroimaging, and neuromodulation that provide converging evidence on symptom-specific brain networks in movement disorders. Although movement disorders have traditionally been conceptualized as disorders of the basal ganglia, cumulative data from brain lesions causing parkinsonism, tremor and dystonia have now demonstrated that this view is incomplete. Several recent studies have shown that lesions causing a given movement disorder occur in heterogeneous brain locations, but disrupt common brain networks, which appear to be specific to each motor phenotype. In addition, findings from structural and functional neuroimaging in movement disorders have demonstrated that brain abnormalities extend far beyond the brain networks associated with the motor symptoms. In fact, neuroimaging findings in each movement disorder are strongly influenced by the constellation of patients' symptoms that also seem to map to specific networks rather than individual anatomical structures or single neurotransmitters. Finally, observations from deep brain stimulation have demonstrated that clinical changes, including both symptom improvement and side effects, are dependent on the modulation of large-scale networks instead of purely local effects of the neuromodulation. Combined, this multimodal evidence suggests that symptoms in movement disorders arise from distinct brain networks, encouraging multimodal imaging studies to better characterize the underlying symptom-specific mechanisms and individually tailor treatment approaches.
© 2024. The Author(s).
Conflict of interest statement
E.G.E, G.M.M., and D.T.C., declare no competing financial or non-financial Interests. V.K. serves as an advisory board member of AbbVie Finland and Nordic Infucare AB, has received travel expenses and speaker honoraria from Abbvie Finland, Nordic Infucare AB and Orion Pharma and research funding from the Finnish Alcohol Research Foundation, the Päivikki and Sakari Sohlberg Foundation, the Finnish Cultural Foundation, the International Parkinson and Movement Disorder Society, and Finnish governmental research funding (VTR). N.P. declares no competing financial interests related to the present article; his contribution to this article reflects entirely and only his own academic expertise on the matter. He has received
Figures

Similar articles
-
Basal ganglia, movement disorders and deep brain stimulation: advances made through non-human primate research.J Neural Transm (Vienna). 2018 Mar;125(3):419-430. doi: 10.1007/s00702-017-1736-5. Epub 2017 Jun 10. J Neural Transm (Vienna). 2018. PMID: 28601961 Free PMC article. Review.
-
Chapter 33: the history of movement disorders.Handb Clin Neurol. 2010;95:501-46. doi: 10.1016/S0072-9752(08)02133-7. Handb Clin Neurol. 2010. PMID: 19892136 Review.
-
Deep Brain Stimulation for Movement Disorders of Basal Ganglia Origin: Restoring Function or Functionality?Neurotherapeutics. 2016 Apr;13(2):264-83. doi: 10.1007/s13311-016-0426-6. Neurotherapeutics. 2016. PMID: 26956115 Free PMC article. Review.
-
Parkinsonism and other movement disorders in 23 cases of neurocysticercosis.Parkinsonism Relat Disord. 2017 Sep;42:47-53. doi: 10.1016/j.parkreldis.2017.06.009. Epub 2017 Jun 20. Parkinsonism Relat Disord. 2017. PMID: 28673613
-
Functional brain networks in movement disorders: recent advances.Curr Opin Neurol. 2012 Aug;25(4):392-401. doi: 10.1097/WCO.0b013e328355aa94. Curr Opin Neurol. 2012. PMID: 22710361 Free PMC article. Review.
Cited by
-
Genetic therapies for movement disorders - current status.J Neurol. 2025 Feb 22;272(3):220. doi: 10.1007/s00415-025-12940-5. J Neurol. 2025. PMID: 39985571 Free PMC article. Review.
-
Advanced Magnetic Resonance Imaging for Early Diagnosis and Monitoring of Movement Disorders.Brain Sci. 2025 Jan 16;15(1):79. doi: 10.3390/brainsci15010079. Brain Sci. 2025. PMID: 39851446 Free PMC article. Review.
References
Publication types
LinkOut - more resources
Full Text Sources

