Chronic Stress Alters Synaptic Inhibition/Excitation Balance of Pyramidal Neurons But Not PV Interneurons in the Infralimbic and Prelimbic Cortices of C57BL/6J Mice
- PMID: 39147579
- PMCID: PMC11351013
- DOI: 10.1523/ENEURO.0053-24.2024
Chronic Stress Alters Synaptic Inhibition/Excitation Balance of Pyramidal Neurons But Not PV Interneurons in the Infralimbic and Prelimbic Cortices of C57BL/6J Mice
Abstract
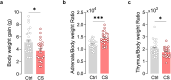
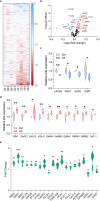
The medial prefrontal cortex (mPFC) plays a pivotal role in regulating working memory, executive function, and self-regulatory behaviors. Dysfunction in the mPFC circuits is a characteristic feature of several neuropsychiatric disorders including schizophrenia, depression, and post-traumatic stress disorder. Chronic stress (CS) is widely recognized as a major triggering factor for the onset of these disorders. Although evidence suggests synaptic dysfunction in mPFC circuits following CS exposure, it remains unclear how different neuronal populations in the infralimbic (IL) and prelimbic (PL) cortices are affected in terms of synaptic inhibition/excitation balance (I/E ratio). Here, using neuroproteomic analysis and whole-cell patch-clamp recordings in pyramidal neurons (PNs) and parvalbumin (PV) interneurons within the PL and IL cortices, we examined the synaptic changes after 21 d of chronic unpredictable stress, in male mice. Our results reveal distinct impacts of CS on PL and IL PNs, resulting in an increased I/E ratio in both subregions but through different mechanisms: CS increases inhibitory synaptic drive in the PL while decreasing excitatory synaptic drive in the IL. Notably, the I/E ratio and excitatory and inhibitory synaptic drive of PV interneurons remained unaffected in both PL and IL circuits following CS exposure. These findings offer novel mechanistic insights into the influence of CS on mPFC circuits and support the hypothesis of stress-induced mPFC hypofunction.
Keywords: I/E ratio; chronic stress; electrophysiology; mPFC; neuroproteomics.
Copyright © 2024 Rodrigues et al.
Conflict of interest statement
The authors declare no competing financial interests.
Figures




References
-
- Amaya KA, Smith KS (2018) Neurobiology of habit formation. Curr Opin Behav Sci 20:145–152. 10.1016/j.cobeha.2018.01.003 - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
