Gastric Adenocarcinoma in Helicobacter pylori-Negative Autoimmune Gastritis: A Case Report and Literature Review
- PMID: 39148949
- PMCID: PMC11326753
- DOI: 10.7759/cureus.66910
Gastric Adenocarcinoma in Helicobacter pylori-Negative Autoimmune Gastritis: A Case Report and Literature Review
Abstract
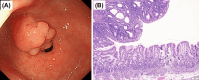
Recent studies have suggested that gastric cancer does not occur in patients with Helicobacter pylori-negative autoimmune gastritis (AIG); however, this notion is controversial. We encountered a case of gastric cancer associated with AIG in which H. pylori infection was excluded. A woman in her 70s was referred to our hospital for endoscopic resection of an antral adenoma. An H. pylori antibodies test, stool antigens test, H. pylori culture, and histological analysis using Giemsa staining yielded negative results. AIG was suspected because the antrum was endoscopically normal but the body was severely atrophic, which are typical findings of AIG. Anti-parietal cell antibodies were 40-fold positive, the gastrin level was 2950 pg/ml, and the pepsinogen I level, pepsinogen II level, and pepsinogen I/II ratio were 6.3 ng/ml, 5.7 ng/ml, and 1.1, respectively. A pathological examination of the gastric body revealed severe oxyntic atrophy with hyperplasia of enterochromaffin-like cells, whereas the antrum showed no pyloric gland atrophy or inflammation. These findings indicated that the patient had H. pylori-negative AIG. Four years later, a depressed lesion in the lower body and a flat lesion at the angle were observed; the former was a poorly cohesive carcinoma, and the latter was a differentiated adenocarcinoma. Surgical resection revealed that the lesion in the lower body was a poorly cohesive carcinoma invading the submucosa with vascular involvement, whereas the lesion in the angle was an intramucosal differentiated adenocarcinoma. A review of previous studies of gastric cancer with H. pylori-negative AIG suggested that patients with histologically and serologically advanced gastritis are at high risk for carcinogenesis. Even in H. pylori-negative cases, severe gastric mucosal atrophy in AIG cases may indicate a carcinogenic risk; therefore, surveillance for gastric cancer is especially recommended for these cases. Large cohort studies on the association between H. pylori-negative AIG and gastric cancer are warranted.
Keywords: autoimmune gastritis; endoscopy; gastric cancer; helicobacter pylori; histology; pepsinogen.
Copyright © 2024, Kishikawa et al.
Conflict of interest statement
Human subjects: Consent was obtained or waived by all participants in this study. Ethics Committee of Tokyo Dental College Ichikawa General Hospital issued approval I-21-43R. Conflicts of interest: In compliance with the ICMJE uniform disclosure form, all authors declare the following: Payment/services info: All authors have declared that no financial support was received from any organization for the submitted work. Financial relationships: All authors have declared that they have no financial relationships at present or within the previous three years with any organizations that might have an interest in the submitted work. Other relationships: All authors have declared that there are no other relationships or activities that could appear to have influenced the submitted work.
Figures





References
-
- Incidence and predictors of gastric neoplastic lesions in corpus-restricted atrophic gastritis: a single-center cohort study. Dilaghi E, Dottori L, Pivetta G, et al. Am J Gastroenterol. 2023;118:2157–2165. - PubMed
-
- Autoimmune gastritis: long-term natural history in naïve Helicobacter pylori-negative patients. Rugge M, Bricca L, Guzzinati S, et al. Gut. 2023;72:30–38. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
